PSA screening alone lacks specificity, leading to substantial overbiopsy and detection of indolent disease. Nomograms—multivariable risk calculators—were developed to move prostate cancer assessment beyond single thresholds. As Drs. Benjamin Tran, Janelle T. West, Soroush Rais-Bahrami, and Kristin K. Porter noted in their ARRS Online Course, the most effective tools integrate clinical variables with mpMRI, making rads central to risk stratification rather than downstream reporters.
Why Nomograms Matter: Traditional PSA-based decision-making treats all patients similarly despite wide variation in underlying risk. Nomograms combine multiple predictors—PSA, prostate volume, digital rectal exam, age, biopsy history, and increasingly PI-RADS assessment—to estimate an individual’s probability of harboring clinically significant prostate cancer (csPCa).
Multiple studies show that nomograms incorporating MRI outperform PSA alone and MRI alone, improving discrimination while safely reducing unnecessary biopsies.
mpMRI’s Role: mpMRI is not just an add-on variable; it is a dominant driver of modern risk models. PI-RADS category, lesion size, and PSA density materially alter predicted risk.
MRI-integrated nomograms consistently achieve AUCs in the 0.80–0.88 range for csPCa detection, while reducing biopsy rates by 20–34% depending on the population and risk threshold chosen. In practical terms, this means fewer low-yield biopsies without a clinically meaningful increase in missed aggressive cancers.
For rads, this elevates the importance of:
- Accurate PI-RADS categorization
- Consistent prostate volume measurement
- Clear reporting of lesion location and size
Small changes in PI-RADS score can significantly shift nomogram output and clinical decision-making.
47-year-old man with history of two negative systematic biopsies at another facility and increasing PSA level (most recently, 35.6 ng/mL). Multiparametric MRI was performed for persistent clinical suspicion of clinically significant prostate cancer
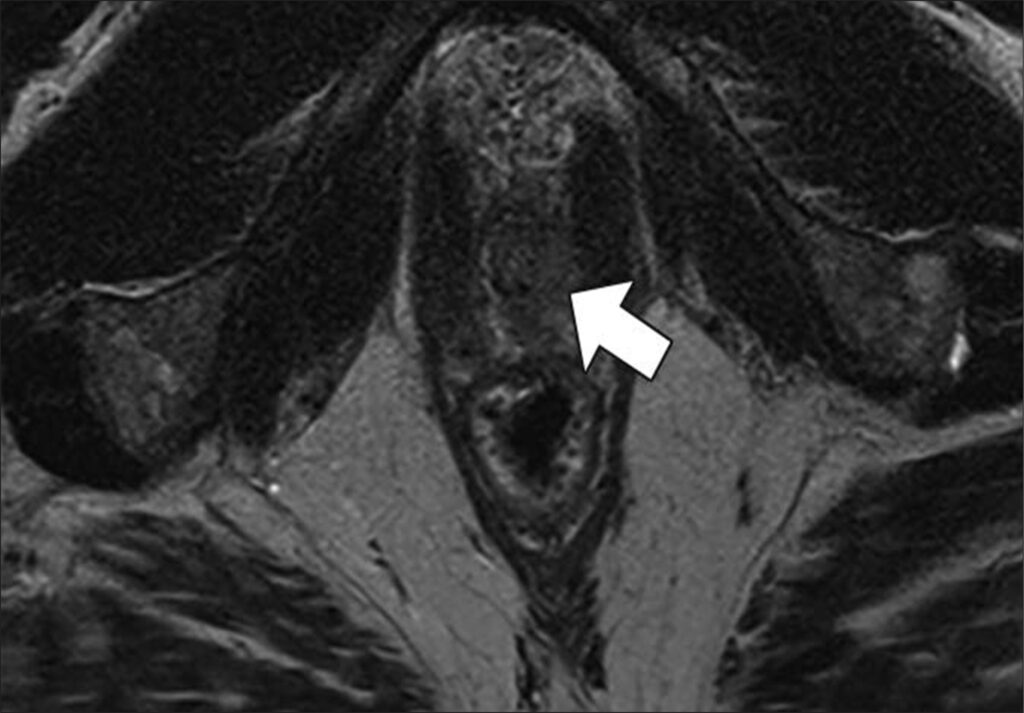
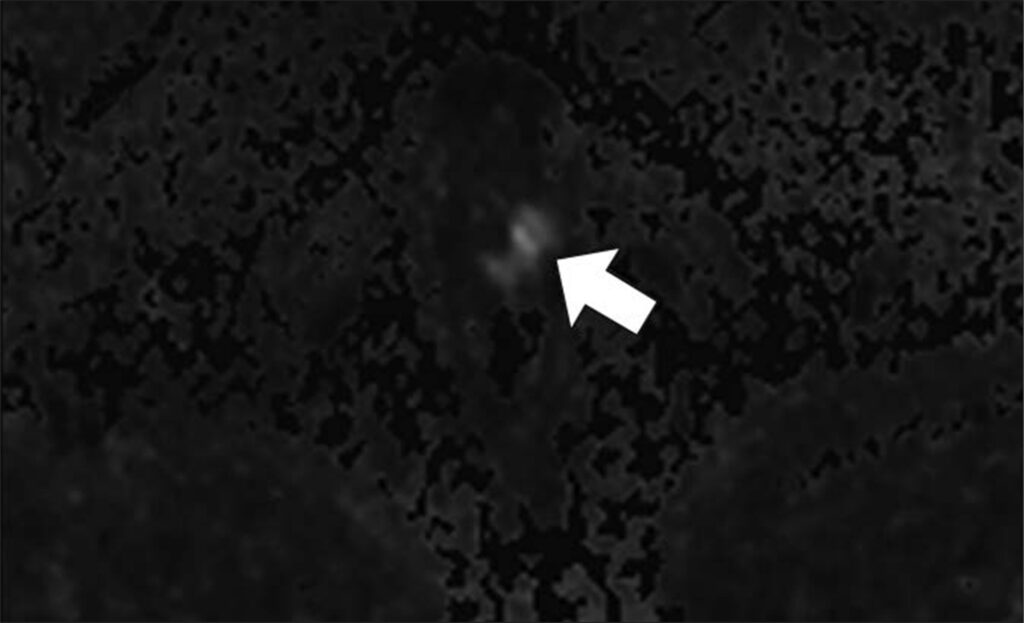
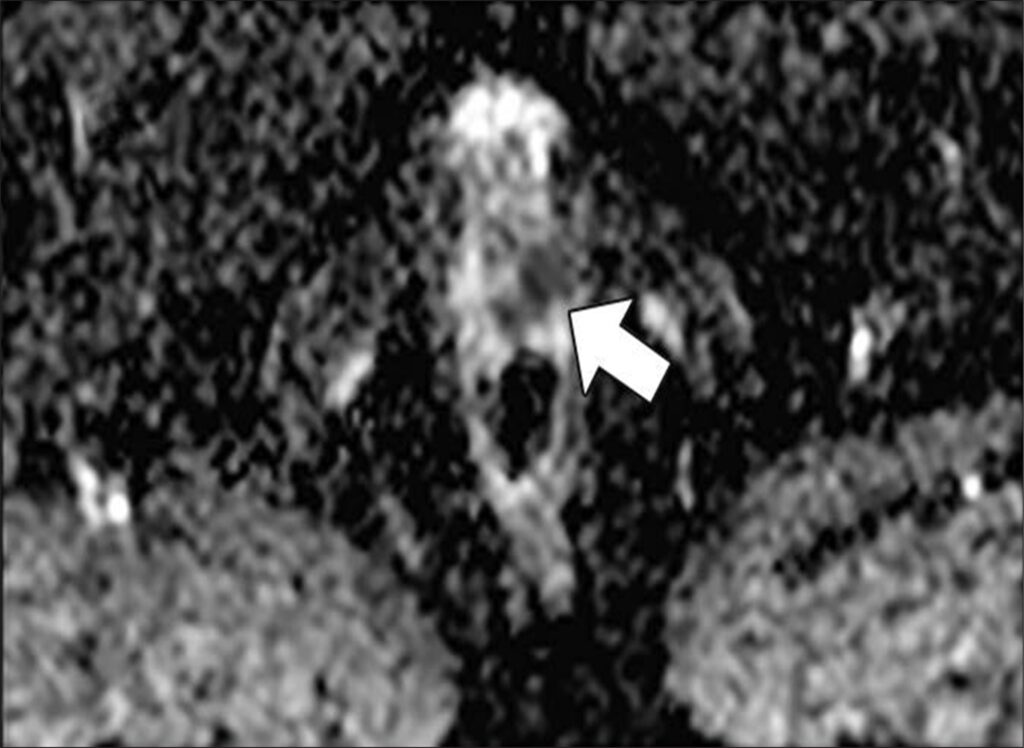
Top to Bottom: Axial T2-weighted (Top), axial high-b-value (2000 s/mm2) (Middle), and calculated ADC (Bottom) MR images show left apical anterior central gland lesion (arrow) with very high suspicion of being clinically significant prostate cancer. Lesion is PI-RADS category 5. MRI–transrectal ultrasound fusion biopsy of lesion resulted in pathologic diagnosis of prostatic adenocarcinoma, Gleason 4 + 3 = 7 (grade group 3). Gleason 4 pattern represents 60% of tumor. Prostate volume calculated with MRI is 40 mL.
PSA Density—A Critical Modifier: PSA density (PSAD) is one of the most powerful adjuncts to PI-RADS. In patients with PI-RADS 1–2 or equivocal PI-RADS 3 lesions, PSAD meaningfully improves negative predictive value—often approaching 90%—and helps identify patients who can safely avoid biopsy.
This is especially relevant in biopsy-naïve men, where avoiding the first biopsy carries the greatest downstream benefit.
Matching Nomogram and Patient: No single nomogram fits every clinical scenario. Some are optimized for biopsy-naïve patients, others for men with prior negative biopsy, and some for mixed cohorts. Understanding these distinctions prevents misapplication and overconfidence in a single risk estimate.
Another important limitation is population dependency. Many widely used nomograms were derived from European or predominantly White cohorts. Without external validation, these tools may overestimate risk in Asian populations and other underrepresented groups. Local calibration and clinical judgment remain essential.
47YO man with two negative systematic biopsies (same patient)
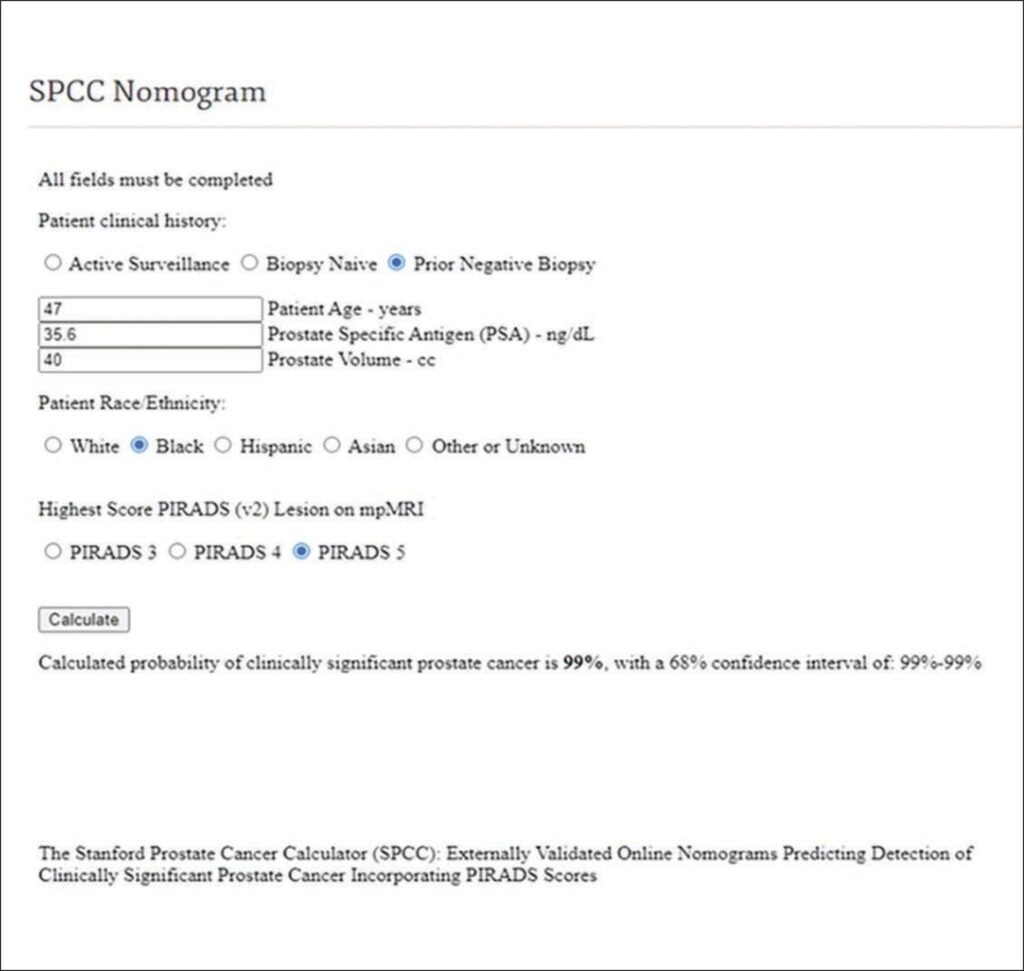
Screenshot of Stanford Prostate Cancer Calculator (SPCC) nomogram calculation shows 99% probability of clinically significant prostate cancer before repeat biopsy. This prediction can be shared with patient during counseling about recommendation for repeat biopsy. Result of repeat MRI– transrectal ultrasound fusion biopsy was consistent with prostatic adenocarcinoma: Gleason 4 + 3 = 7 (grade group 3). Gleason 4 pattern represented 60% of tumor.
What Does This Mean for Rads? Nomograms reposition rads as active participants in prostate cancer decision-making. High-quality MRI interpretation directly influences whether a patient undergoes biopsy, surveillance, or reassurance.
In multidisciplinary care, radiologists who understand nomogram inputs and limitations can:
- Explain risk estimates to urologists with confidence
- Support biopsy deferral when appropriate
- Improve alignment between imaging findings and management
| Nomogram / Calculator | Best Use Case | Key Inputs | Strengths | Limitations |
| MRI-ERSPC RC3 / RC4 | Biopsy-naïve patients | PSA, PSAD, DRE, prostate volume, PI-RADS | Strong external validation; good balance of biopsy reduction and csPCa detection | Derived largely from European cohorts |
| ModRad | Biopsy-naïve patients | PSA, PSAD, PI-RADS, clinical variables | High discrimination; MRI-forward design | Less validated in prior biopsy populations |
| ModDis | Prior negative biopsy | PSA, PSAD, PI-RADS, biopsy history | Reduces repeat biopsies while maintaining csPCa sensitivity | Less applicable to biopsy-naïve patients |
| Stanford Prostate Cancer Calculator | Mixed populations | PSA, DRE, biopsy history, MRI findings | Broad applicability; flexible inputs | Slightly lower performance than MRI-specific models |
| PCPT Risk Calculator | Legacy / screening context | PSA, age, race, DRE | Historically influential | Does not incorporate MRI; lower specificity |
| ERSPC (non-MRI) | Screening populations | PSA, DRE, prostate volume | Well studied | Outperformed by MRI-integrated tools |
Bottom Line: mpMRI-integrated nomograms transform prostate cancer assessment from threshold-based screening to personalized risk prediction. Radiologists who deliver precise PI-RADS scoring and understand how those scores drive nomograms add measurable, patient-level value.

Leave a Reply