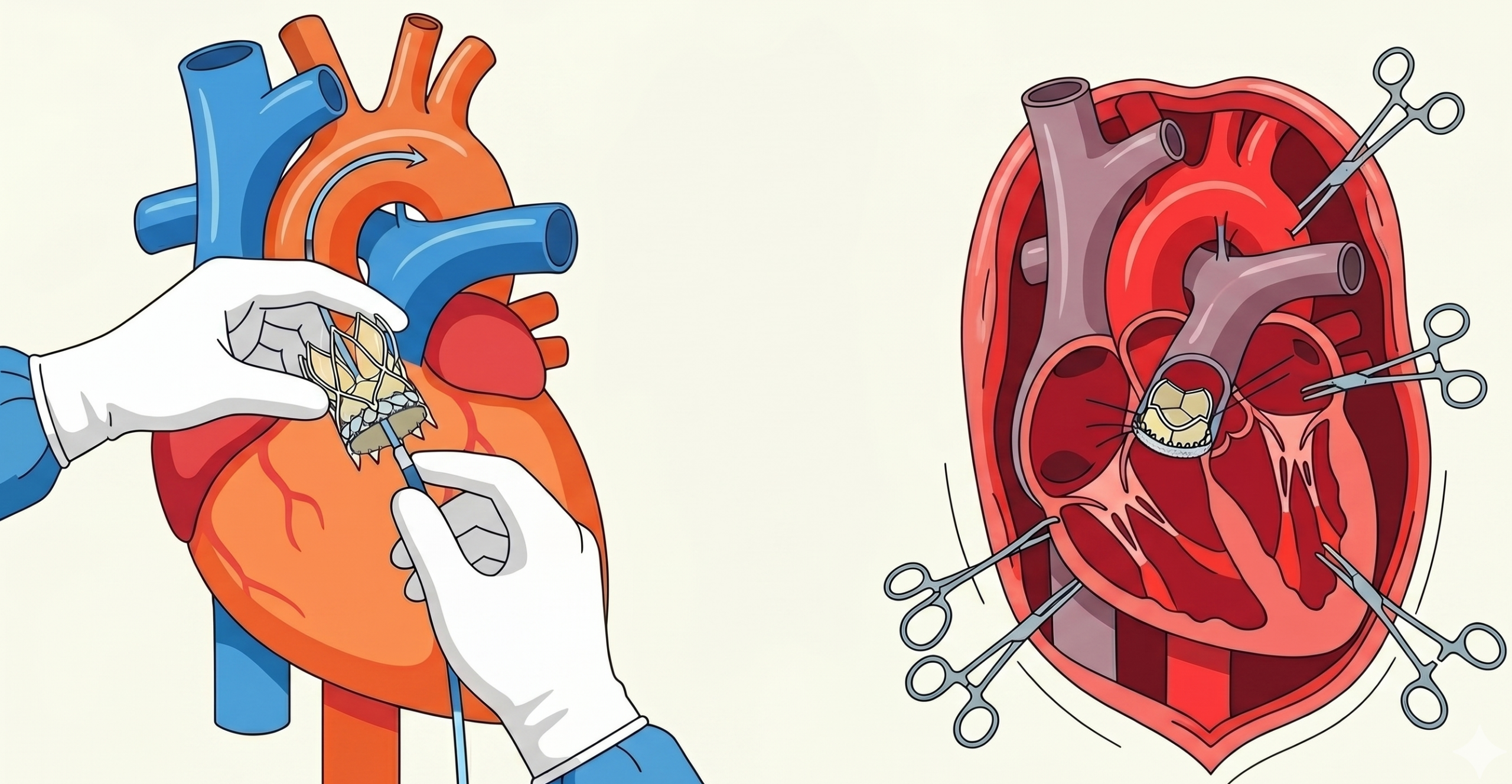
For patients with severe aortic stenosis and a life expectancy greater than one year, the choice between transcatheter aortic valve replacement (TAVR) and surgical aortic valve replacement (SAVR) is a complex decision made by a multidisciplinary heart team.
Three Things: Although TAVR was initially the standard for patients with high surgical risk, its use is rapidly increasing. Today, TAVR’s approved for low-risk patients, too. However, as noted by Matthew Stib, MD, of Mayo Clinic Arizona during the ARRS Online Course Mastering CT for TAVR and TMVR, the choice still hinges on three primary features: age, surgical risk, and life expectancy.
- <65 years: SAVR is preferable. Surgical valves have established longevity, whereas TAVR has only been in use for about a decade, and its long-term durability is less certain.
- 65–80 years: Both are viable options; the choice depends on patient preference and specific clinical or anatomical factors.
- >80 years: TAVR is the preferred approach.
- High Surgical Risk: TAVR is favored, regardless of age.
Two Tips: Pre-procedural CT remains the reference standard for TAVR planning, serving two critical roles: sizing the prosthesis at the aortic root and evaluating vascular access in the thorax, abdomen, and pelvis.
Beyond the Valve: Recent research indicates that rads should look at more than just the valve to predict patient outcomes. Features of right heart failure identified on pre-procedure CT are independent predictors of 1-year mortality after TAVR:
- Pulmonary Artery Dilation: A median main size of 3.2 cm was associated with death within 1 year, compared to 2.9 cm in survivors.
- Pericardial Effusion: The presence of a moderate-to-large pericardial effusion is a strong predictor of mortality, even when adjusting for the Society of Thoracic Surgeons clinical risk score.
Bottom Line: As TAVR becomes more common, the rad’s role is expanding from procedural planning to risk stratification. Identifying signs of right heart failure—like a dilated PA or pericardial effusion—can help identify high-risk candidates who may have poor outcomes, despite a technically successful procedure.