Published January 4, 2021

Ian Y. M. Chan
Department of Medical Imaging
Western University

Tyler Cossetto
Department of Medical Imaging
Western University

Bradley Davis
Department of Medical Imaging
Western University

Victor Lam
Department of Medical Imaging
Western University

Timo Krings
Division of Neuroradiology
University of Toronto
The coronavirus disease (COVID-19) pandemic is now the most significant pandemic in over a century. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19, affects multiple body systems by binding to the angiotensin-converting enzyme 2 (ACE2) receptors on cells. Although pulmonary manifestations of COVID-19 have garnered much attention, its neurological impact can be as devastating. Neurological manifestations include ischemic stroke, intracranial hemorrhage, encephalopathy, and peripheral neuropathy. Neurological symptoms are common and include headache, dizziness, myalgia, seizures, weakness, strokes, and alterations of consciousness. Although rarely isolated, disorders of smell and taste have also been seen in COVID-19. For some patients, cognitive symptoms have been found to last for months following infection. Radiologists must be attuned the neurological manifestations of COVID-19, as early identification of these findings may help to guide management and reduce disease transmission.
Ischemic Stroke
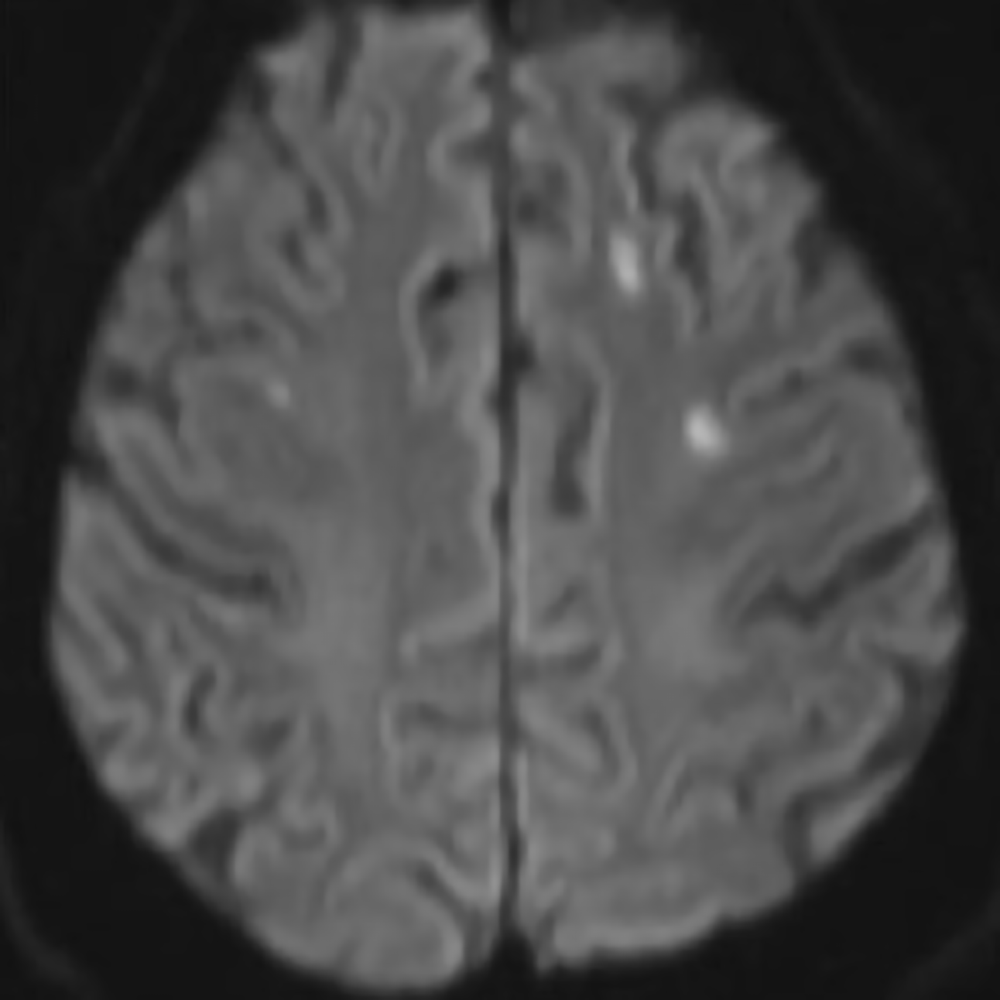
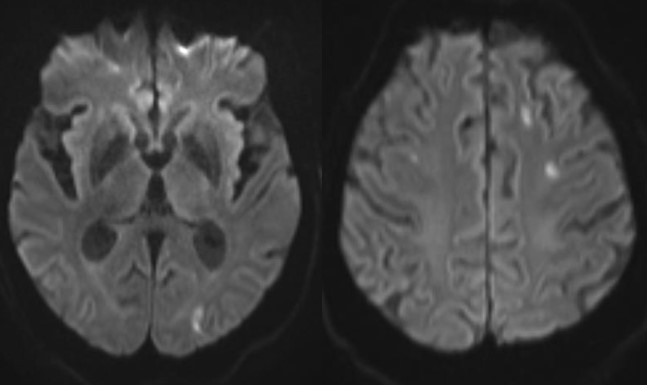
The incidence of ischemic stroke associated with COVID-19 in hospitalized patients is between 0.4% and 2.7% but may vary with infection severity. The association between ischemic stroke and COVID-19 has been proposed since the outset of the outbreak, fueled by the rationale that infectious processes like COVID-19 are prothrombotic through an endotheliitis-mediated mechanism [Fig. 1].
Fig. 1—74-year-old male patient with COVID-19 and no comorbidities who presented with acute confusion. Brain MRI with axial DWI images demonstrates multiple foci of acute ischemia in the deep white matter that are atypical for embolic lesion given their subcortical location. Extensive stroke etiology workup was negative without cardiac or arterial embolic source. This microangiopathic ischemia was linked to hypercoagulability and small vessel occlusion related to COVID-19.
Initial concern was raised following an increased incidence in large vessel occlusion (LVO) strokes. In April 2020, an early case series published in the New England Journal of Medicine highlighted five cases with LVO stroke in New York City and suggested that LVOs may be a presenting feature of COVID-19 in young adults. A case-control study from New York City published in AJR showed that LVOs were present in 32% of code stroke patients with COVID-19, compared to 15% without COVID-19. Recently, a systematic review by Canadian researchers confirmed that 1.5% of patients admitted to hospitals with COVID-19 suffered an ischemic stroke with a resultant mortality of 35% for all stroke types. More concerning, in patients younger than 50, half had no other symptoms of infection at the time of ischemic stroke onset, and 69% presented with LVO.
By contrast, 17 stroke centers in Europe and North America reported a 32% reduction in endovascular thrombectomy (EVT) procedures during the pandemic. The number of patients undergoing neuroimaging for stroke in the United States also decreased by 39%. Proposed reasons for this discrepancy include reluctance of patients to seek care due to fear of contracting COVID-19 and an overwhelmed health care system. With this divergent data, strong epidemiological studies are needed to determine the underlying reasons for these statistics.
Intracranial Hemorrhage
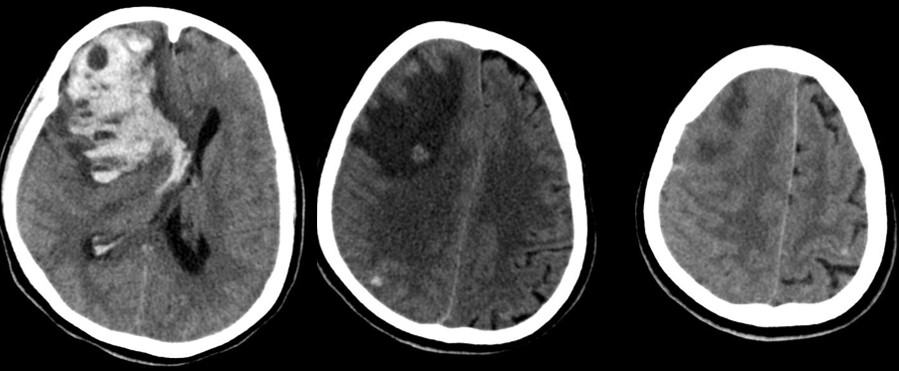
A growing number of studies have investigated the relationship between COVID-19 and intracranial hemorrhage (ICH). The incidence of ICH in COVID-19 ranges from 0.2% to 0.9%. ICH has a much higher likelihood of occurring in hospitalized patients with COVID-19 who receive anticoagulation or extracorporeal membrane oxygenation.
Intracerebral and subarachnoid hemorrhage, as well as hemorrhage from cerebral venous thrombosis, have been reported in COVID-19. The majority of intracerebral hemorrhages are lobar with deep structure and infratentorial hemorrhages being less common. Microhemorrhages in the juxtacortical white matter and corpus callosum (particularly the splenium), multilobar microhemorrhages, and single large lobar hemorrhages have been observed.
At this time, it is unclear if ICH seen with COVID-19 is causative or coincidental. Two prominent hypotheses suggesting causation point to the pathophysiology of sepsis and the activation of the renin-angiotensin system leading to endovascular disruption. A retrospective case series, which suggested these findings are coincidental, showed that most patients with ICH and COVID-19 could be attributed to prior trauma and known suspected hemorrhage before contracting the virus. This highlights the challenge in accurately attributing ICH events to COVID-19 [Fig. 2].
Encephalopathy
Encephalopathies have gained increasing attention as unusual manifestations of COVID-19 and harbingers of more severe disease. Similar coronavirus disease, such as severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), have shown a similar propensity to cause acute encephalopathy. The prevalence of this condition ranges from 0.04% to 0.2% for COVID-19 patients. Acute encephalopathy was associated with more severe infection and worse clinical outcomes in hospitalized patients, which makes identification important.
The etiology of acute encephalopathy associated with COVID-19 is not entirely clear. Cases have been reported in the literature. Meningoencephalitis was shown in a young adult patient without significant respiratory disease. Leukoencephalopathy and acute hemorrhagic necrotizing encephalopathy were noted in case reports. Encephalitis was also seen in an adolescent patient with severe infection.
The primary imaging finding of encephalopathy was nonspecific cortical T2/FLAIR hyperintensities with associated restricted diffusion. Bilateral FLAIR hyperintense thalamic lesions have been characteristically described in acute necrotizing encephalopathy. Imaging features of posterior reversible encephalopathy syndrome and hypoxic-ischemic encephalopathy, as well as exacerbation of demyelinating disease, were also seen. Kihira et al. described multiple cases of white matter disease related to hypoxic leukoencephalopathy, acute disseminated encephalomyelitis, and direct viral encephalitis attributed to COVID-19. A serious condition linked with COVID-19 known as multisystem inflammatory syndrome in children (MIS-C) can cause neurocognitive symptoms, and in four cases, MRI revealed signal abnormalities in the splenium. Consequently, radiologists must have a high index of suspicion for a variety of encephalopathies in patients with COVID-19.
Peripheral Neuropathy
Various studies have identified peripheral neuropathy related to COVID-19. Indeed, since ACE2 receptors are found in the olfactory epithelium, the neuroinvasive potential of SARS-CoV-2 has been a topic of investigation.
Anosmia may be the most common neurological symptom. Inflammatory obstruction of the olfactory clefts is known to impair olfaction. CT and MR images in patients with COVID-19 have shown obstruction of the olfactory clefts, which has been thought to lead to anosmia. Imaging of infected patients has also shown olfactory bulb atrophy and MRI signal changes that can resolve on follow-up imaging.
Although rare, Guillain-Barré syndrome (GBS) has also been reported in COVID-19. Comprising a heterogeneous group of immune-mediated polyneuropathies, GBS classically presents as rapid symmetrical ascending muscle paresis or paralysis. To date, at least 73 cases of GBS have been reported in patients with COVID-19. GBS associated with COVID-19 has been shown to exhibit asymmetrical thickening and hyperintensity of the post ganglionic roots supplying the brachial and lumbar plexuses on STIR images. Facial nerve, nerve root, and leptomeningeal enhancement have been reported. Of note, MRIs of the brain and spine in these patients are most often normal. Other neuromuscular conditions, such as generalized myoclonus and rhabdomyolysis, have also been rarely reported.
We have highlighted a number of important neurological manifestations of COVID-19. Our understanding of this novel coronavirus and its neurological manifestations continues to improve, and the epidemiology between these conditions and COVID-19 are frequently being refined. We remain hopeful that the management and prevention of COVID-19 will continue to advance with the development of new therapeutics and vaccines in the coming months and beyond. As radiologists, we should be cognizant of the fact that imaging has crucial role to play in diagnosing many of these neurological manifestations. Moreover, long-term sequelae of these conditions will often necessitate further follow-up imaging, ensuring our continued involvement in patient care for the foreseeable future.