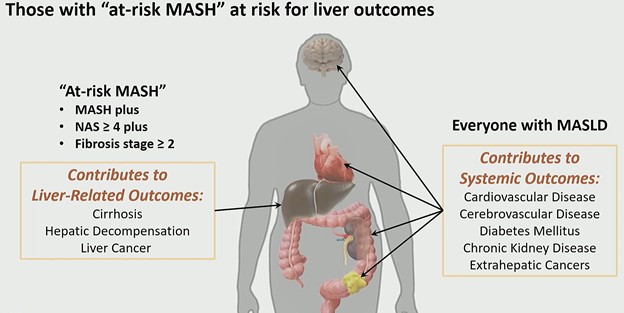
A new term is reshaping hepatology and radiology alike: at-risk MASH. As described by Claude B. Sirlin, MD, during the ARRS QuickByte “Latest Insights Into MASLD,” this subgroup represents patients with metabolic dysfunction–associated steatohepatitis (MASH) who have a nonalcoholic fatty liver disease (NAFLD) activity score ≥4 and fibrosis stage ≥2—the cohort most likely to experience liver-related outcomes and qualify for pharmacologic therapy.
Why “At-Risk” Changes the Conversation: MASLD progresses along a biologic continuum:
- Steatosis appears first and increases early.
- Inflammation and ballooning signal transition to MASH.
- Fibrosis stages 1–4 define structural progression.
Critically, as fibrosis advances, steatosis may decline—and in end-stage cirrhosis, fat may disappear entirely. This has major imaging implications. Proton density fat fraction (PDFF) is an excellent marker of steatosis and often correlates with MASH. But PDFF is not necessarily the best marker of at-risk MASH, because patients with significant fibrosis (stage ≥2) may demonstrate decreasing fat content.
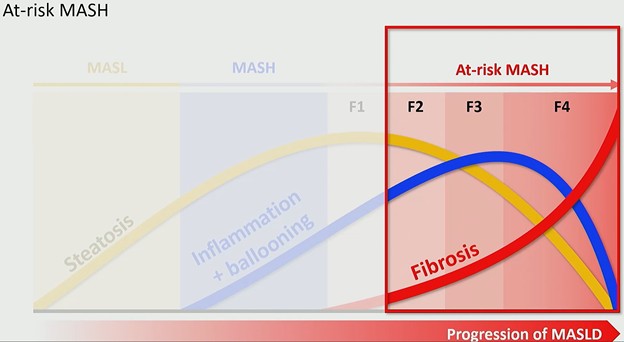
In other words: less fat does not mean less risk.
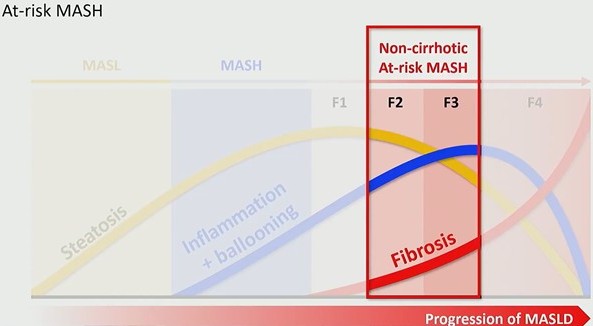
A Therapeutic Threshold: The FDA approval of Resmetirom on March 14, 2024 marked the first pharmacologic therapy for MASH. However, this therapy is not broadly indicated. At approximately $50,000 per year, it is targeted specifically to patients with non-cirrhotic at-risk MASH (i.e., those with active disease and stage ≥2 fibrosis, but not cirrhosis). This shifts the rad’s role from descriptive to decisional. Identification now determines treatment eligibility.
The Quantitative Imaging Era: Eligibility criteria for Resmetirom were MRI-based, placing quantitative MR biomarkers at the center of clinical decision-making. This marks a broader transition: Radiology is moving from qualitative pattern recognition to measurable disease phenotyping. While MRI has led this evolution, ultrasound-based quantitative biomarkers are expected to follow, expanding access and scalability in the near term. Recently, Dr. Sirlin and Scott B. Reeder, MD, emphasized this shift in an editorial introducing rads to the expanding role of quantitative MRI biomarkers in metabolic liver disease.
Pattern → Risk → Responsibility: MASLD progresses from fat accumulation to inflammation and fibrosis. → Patients with MASH plus stage ≥2 fibrosis face liver-related outcomes and are candidates for therapy. → Rads must accurately stage fibrosis and quantify disease to guide referral and treatment.
Clinical Takeaway: The most important insight is conceptual . . . At-risk MASH is a fibrosis-driven definition, not a fat-driven one.
Quantitative imaging, particularly MR-based biomarkers, now determines:
- Who gets referred
- Who receives therapy
- Who remains under surveillance
This is not simply a nomenclature update. It is a reimbursement, therapeutic, and prognostic pivot point for abdominal imaging.
Bottom Line: In the era of approved therapy, identifying at-risk MASH is no longer optional precision; it is actionable medicine.

Leave a Reply