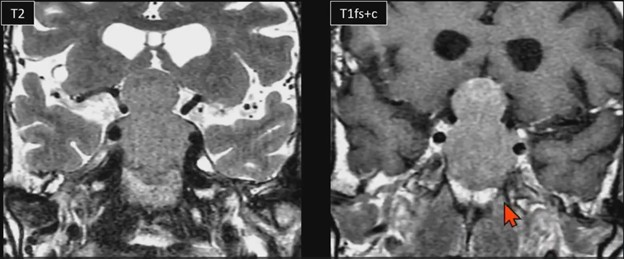
Do you wanna see a snowman? University of Mississippi neuroradiologist Charlotte S. Taylor, MD, illustrates a specific abnormality of the sella/suprasellar region in this ARRS Web Lecture—now available in your library.
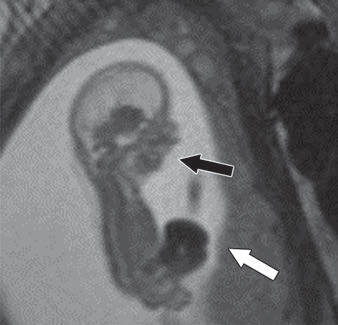
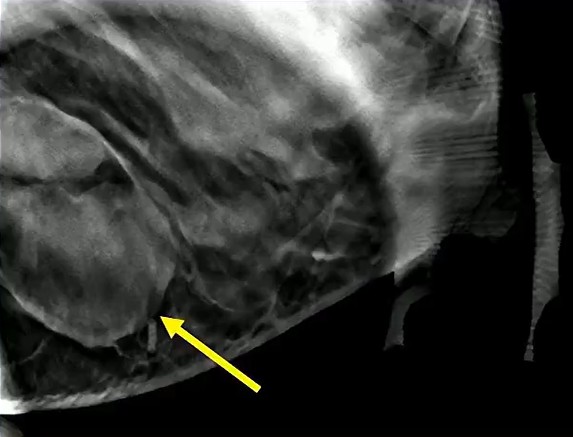
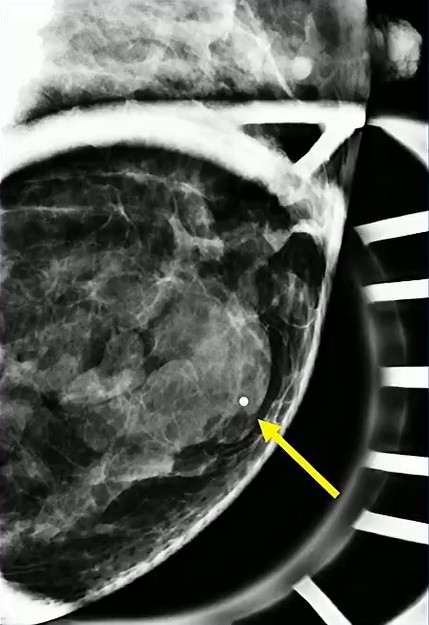
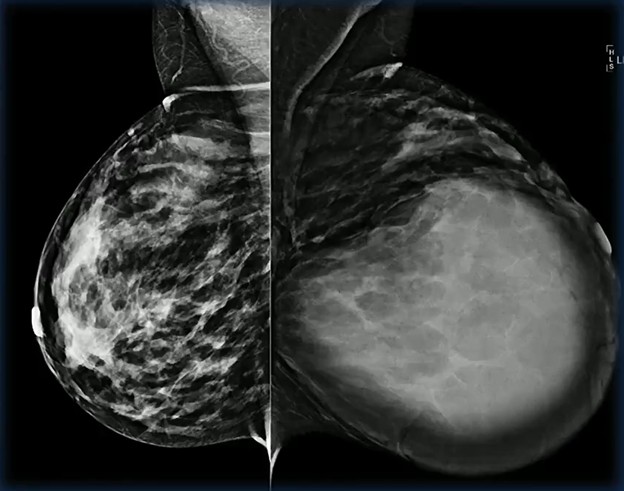
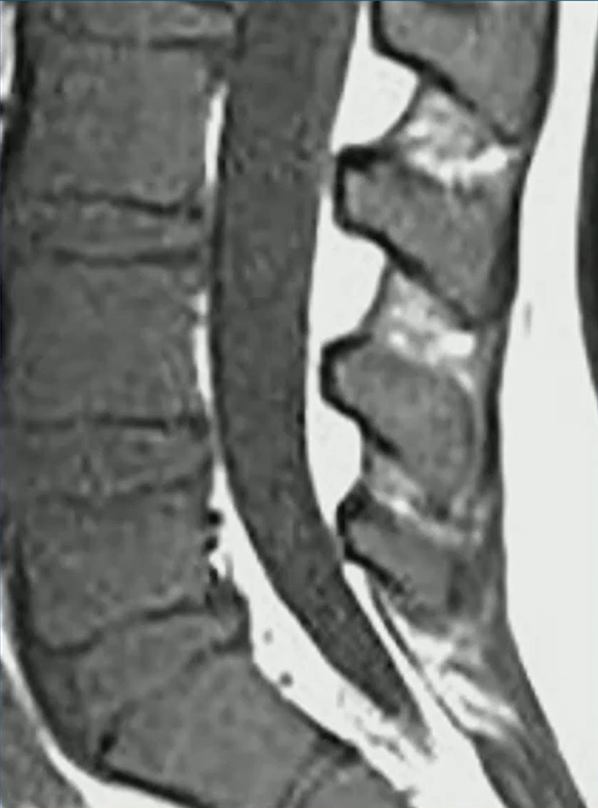
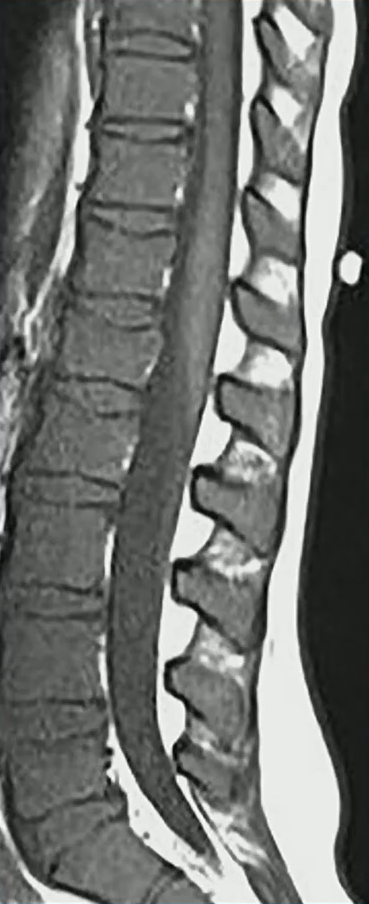
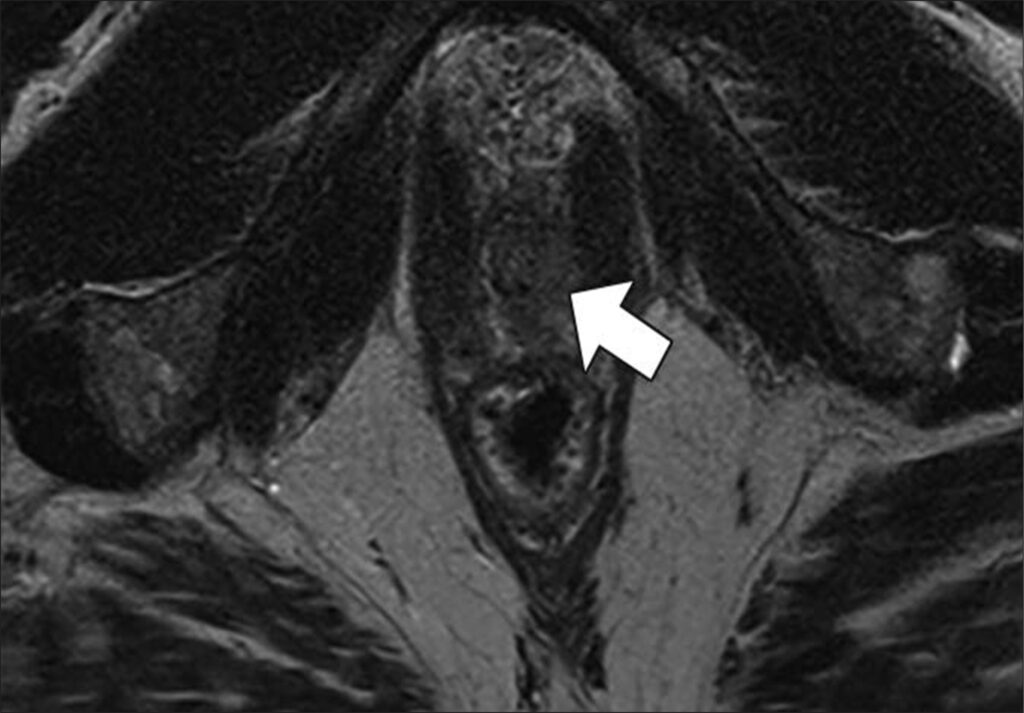
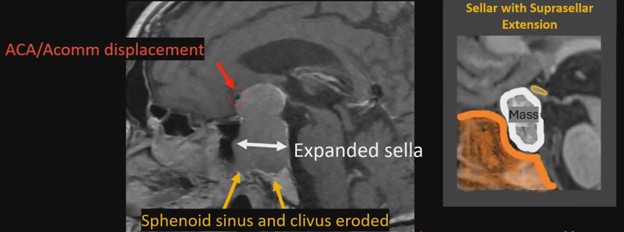
Waist Deep: When viewing a coronal or sagittal MRI of a classic pituitary macroadenoma (PM), you’ll often spot a snowman-shaped, homogeneously enhancing mass. This characteristic shape occurs due to a sella mass that develops a suprasellar extension, creating a “waist” as it pushes upward. In these pronounced cases, the normal pituitary tissue is usually completely displaced or obscured. You may also notice local bony involvement, including erosions in the sphenoid sinus and clivus.
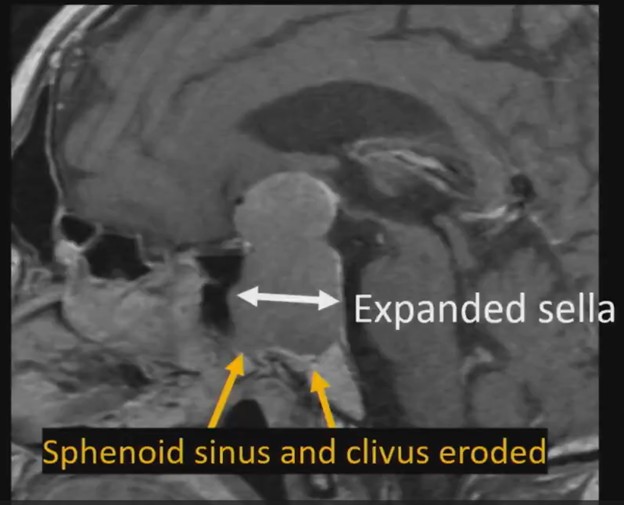
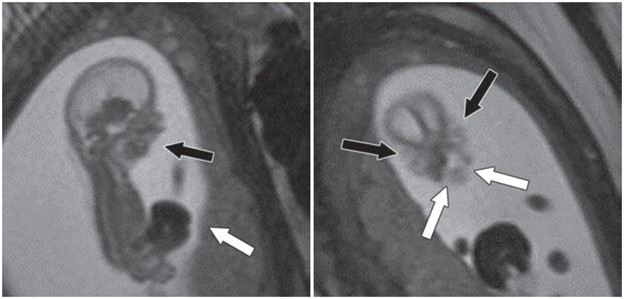
Hat Trick: As the tumor extends superiorly, it exerts mass effect on the optic chiasm. The chiasm can become so severely stretched that it is difficult to visualize directly—effectively forming a sadly stretched “hat” resting atop our snowman’s noggin.
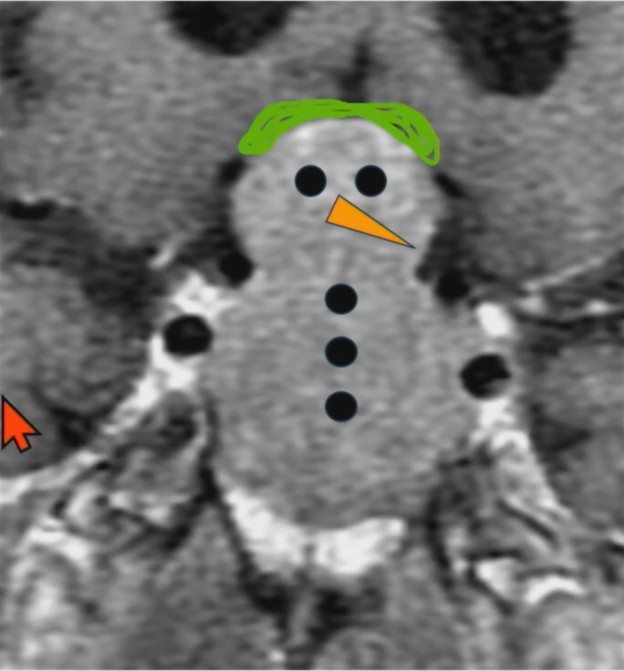
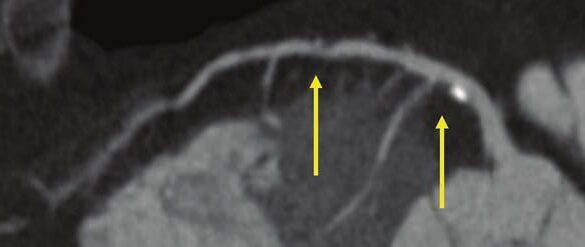
If the optic chiasm is too stretched to be seen clearly, look for the anterior cerebral arteries (ACA) and the anterior communicating arteries (Acomms). Because these vessels supply perforators to the optic chiasm, they are physically tethered to it. As these vessels are displaced superiorly by the mass, you can use ACA/Acomms as a reliable surrogate for location.
What About BHA? By definition, a PM measures 10 mm or greater in size. The non-functional variants are the most common and typically present due to visual disturbances resulting from the suprasellar mass effect. Whereas rads have been taught that pituitary adenomas cause true bitemporal hemianopsia (BHA), complete BHA is exceedingly rare, occurring in less than 1% of patients. Instead, incomplete bitemporal or mixed visual field defects are the most common presentation, seen in about 42.6% of patients. Notably, these visual field defects typically only occur when the tumor displaces the optic pathway by 3 mm or more.
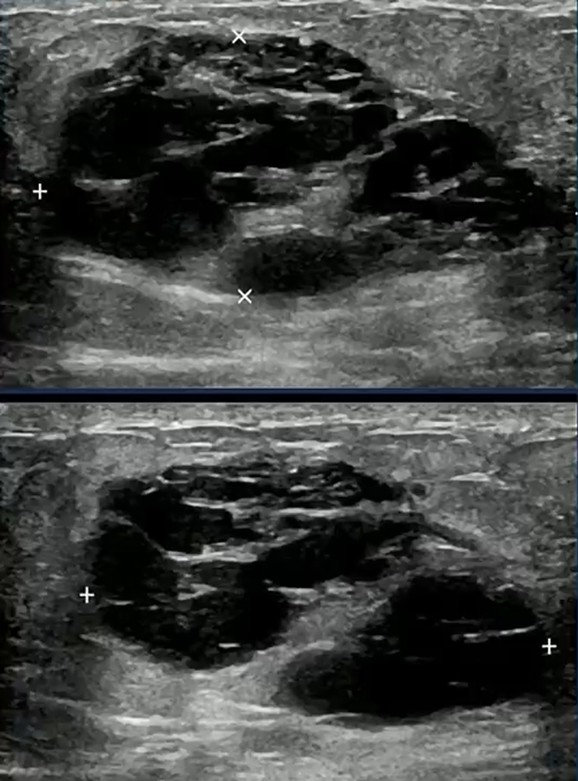
Surgical Insight: Treatment for these lesions is generally surgical—most frequently an endoscopic transsphenoidal approach—with the notable exception of prolactinomas, which are treated medically using dopamine agonists. For surgical candidates, preoperative MRI features can help predict procedural success. Results from AJR show tumors with macrocysts, macrohemorrhage, or enhanced diffusivity (ADC ratio > 1.1) are highly resectable via transsphenoidal approach. Conversely, solid tumors with restricted diffusion tend to have a higher reticulin content, making them more rigid and more likely to fail transsphenoidal resection, perhaps requiring transcranial surgery.
Surveilling a Nonresected PM: For conservative patient management, the debate remains. Some experts argue gadolinium-based contrast agents are essential to assess tumor invasiveness, detect solid components, and evaluate markers of aggressive proliferation. And others counter that an unenhanced coronal T2-weighted MRI is perfectly adequate for tracking dimensions and optic compression, allowing your patient to avoid the costs, discomfort, and long-term retention risks associated with IV contrast.