Published March 8, 2023

Andrew L. Callen
@AndrewCallenMD
Department of Radiology, Neuroradiology Section, University of Colorado Anschutz Medical Campus
Spontaneous intracranial hypotension (SIH) is a disabling headache disorder caused by abnormal leakage of CSF through a dural defect, ruptured meningeal diverticulum, or CSF venous fistula (CVF) [1]. The clinical hallmark of this syndrome is a headache that improves when supine and worsens when upright. Although iatrogenic post–dural puncture headaches have been recognized for over a century, the understanding of spontaneous leakage of CSF and its associated syndrome has evolved rapidly over the past decade [2]. CVF in particular was first recognized in 2014, so our understanding of how to localize and treat this specific pathology is in its relative infancy [3].
As radiologists, we find ourselves uniquely positioned to both diagnose and treat patients with spontaneous intracranial hypotension (SIH). Understanding the spectrum of SIH imaging abnormalities in the brain and spine, how to implement specialized MRI and myelographic protocols to localize CSF leaks and CVF, and the treatment options available allows optimal care of these patients [4].
First-Line Evaluation With Noninvasive Imaging
MRI of the Brain
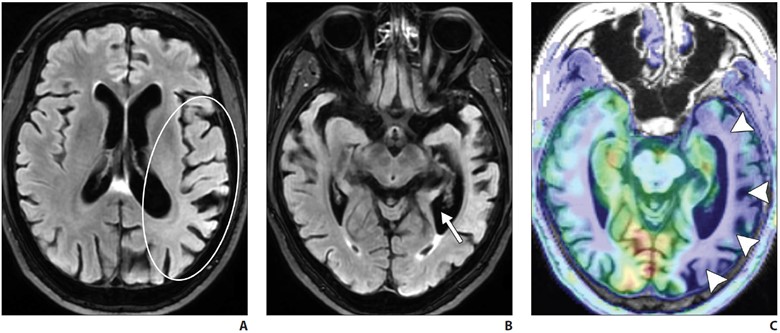
Routine MRI brain protocols consisting of contrast-enhanced 3D T1-weighted, susceptibility-weighted, and T2-weighted FLAIR sequences are sufficient to evaluate evidence of underlying SIH. Changes during SIH can be framed in terms of the Monro-Kellie doctrine: in normal states, the volumes of blood, CSF, and brain parenchyma are in equilibrium [5]. In response to abnormal CSF deliquoration, compensatory enlargement or deformation of brain parenchyma and vascular structures occurs, resulting in sagging of the brainstem, diffuse circumferential pachymeningeal thickening, epidural venous engorgement, and pituitary hyperemia [6]. Subdural hygromas or hematomas may occur secondary to distention of intracranial venous structures, which must not be mistaken for posttraumatic hemorrhage, as surgical evacuation of the collections will not improve the patient’s underlying pathology [6]. Hemosiderosis, particularly of the infratentorial brain, can be seen and may be due to repeated microtrauma to the epidural venous plexus at the dural defect, resulting in circulation of subarachnoid blood products [7] (Fig. 1).
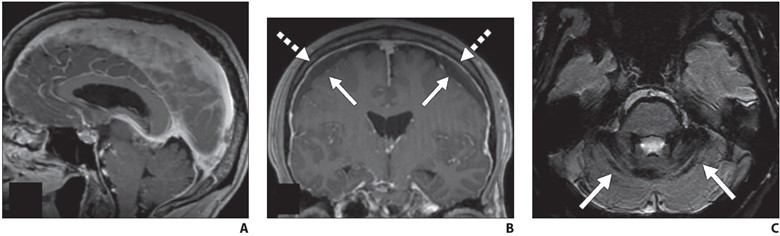
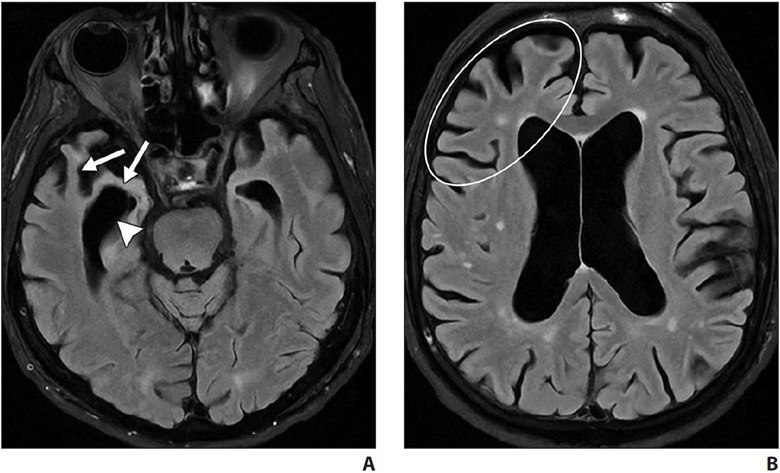
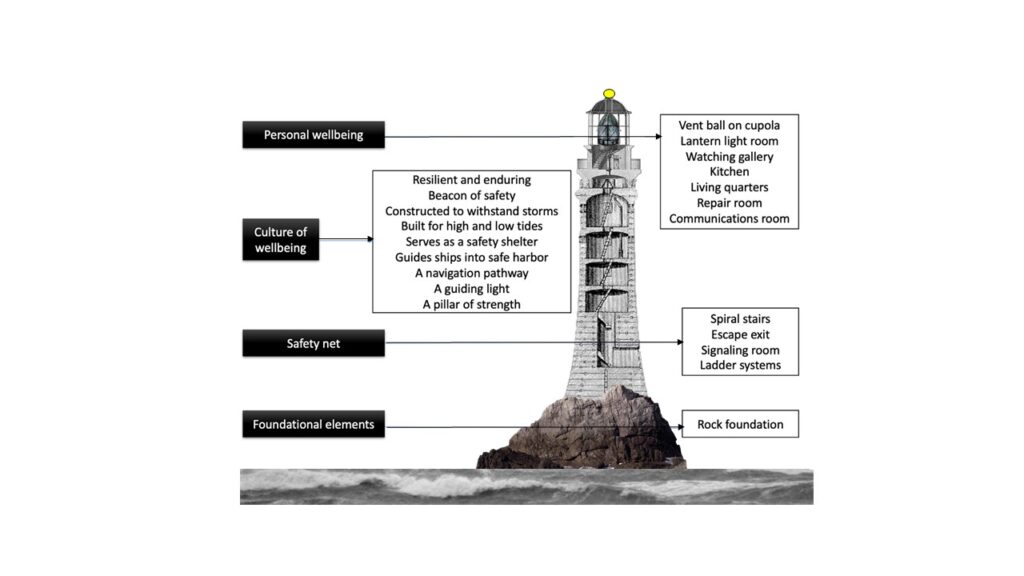
Fig. 1—MRI findings of spontaneous intracranial hypotension.
A, Sagittal contrast-enhanced T1-weighted image shows sagging of brainstem with effacement of mamillopontine, prepontine, and suprasellar intervals as well as engorgement of dural venous sinuses.
B, Coronal contrast-enhanced T1-weighted image shows bilateral subdural hygromas (solid arrows) and diffuse smooth pachymeningeal thickening and enhancement (dashed arrows).
C, Axial susceptibility-weighted image shows diffuse infratentorial hemosiderosis (arrows).
Emerging evidence suggests that although findings of SIH are specific for an underlying CSF leak, brain MRI may have limited sensitivity for these findings, particularly in chronic leaks [8]. A recent meta-analysis suggests that at least 19% of patients with proven CSF leaks may have normal findings on brain MRI [9]. Additional studies have reported that as a CSF leak persists, imaging findings either resolve or become more subtle, particularly diffuse pachymeningeal enhancement [10].
The following Bern criteria [11] have been developed as a scoring system using both qualitative and quantitative information on brain MRI to assign high, intermediate, or low probability of SIH:
Bern Criteria for Assessment of Spinal CSF Leak Based on Brain MRI Findings
| MRI Finding | No. of Points |
| Pachymeningeal enhancement | 2 |
| Venous engorgement | 2 |
| Suprasellar effacement (≤ 4 mm) | 2 |
| Subdural collection | 1 |
| Prepontine effacement (≤ 5 mm) | 1 |
| Mamillopontine effacement (≤ 6.5 mm) | 1 |
Standardized reporting for brain MRI with clinical indications suggestive of underlying SIH can appropriately frame the risks and benefits of further invasive diagnostic testing and avoid wrongly characterizing a patient as not having SIH because of a negative brain MRI examination. The radiologist’s understanding of limited sensitivity of brain MRI is critical for patients with SIH; it is strongly encouraged to adopt language that conveys probabilities of underlying SIH and incorporation of Bern scoring to not prevent further diagnostic workup in cases of high clinical suspicion.
MRI of the Spine
If SIH is suspected on the basis of clinical or imaging findings, imaging evaluation of the spine must be performed, as most CSF leaks resulting in SIH occur in the spine, not from the skull base [12]. In the upright position, intracranial pressure is slightly negative relative to atmospheric pressure, and pressure in the spinal compartment is slightly positive, which can result in spontaneous leakage of CSF in sufficient quantities to result in SIH [13]. This phenomenon is critical for the radiologist to understand, as it is not rare for patients to be inappropriately referred for CT cisternography in the context of orthostatic headaches, positive brain MRI, and rhinorrhea for suspicion of an underlying skull base leak.
Although routine spine imaging protocols can adequately delineate a dorsal or ventral spinal epidural CSF collection, leaks originating from the lateral dura (ruptured nerve root axilla or meningeal diverticulum) will not be evident on routine axial non–fat-saturated T2-weighted MRI. Similarly, laterally directed meningeal diverticula will not be completely characterized on routine protocols (Fig. 2).
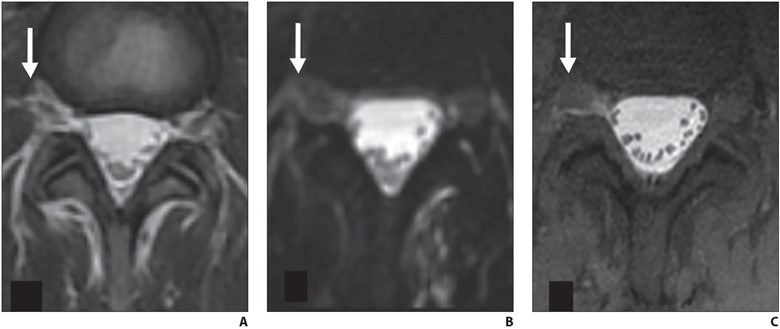
Fig. 2—Axial lumbar spine MRI findings of 39-year-old woman with orthostatic headache. (B and C adapted from [4])
A and B, Axial T2-weighted (A) and heavily T2-weighted fat-saturated (B) images do not delineate any clear extradural fluid. T2-weighted hyperintensity lateral to dura (arrow, A) blends with fat and epidural veins (A). Three-dimensional CSF leak protocol shows minimal T2-weighted asymmetry (arrow, B) in right L4–5 neural foramen at same level (B).
C, Delayed T1-weighted fat-saturated image with intrathecal gadolinium confirms extradural CSF (arrow) at this level. Patient was treated with transforaminal epidural fibrin patch that relieved symptoms.
Therefore, specialized 3D heavily T2-weighted fat-saturated spine MRI protocols should be used routinely in the workup of potential underlying SIH. At my institution, we use a Siemens Healthcare protocol with coronal fat-saturated HASTE images with 256 echo train length, 0.9-mm3 isotropic resolution, and TR/TE of 8000/271. These images are reformatted in sagittal and axial planes, reformatted into a coronal volumetric maximum intensity projection, and supplemented with sagittal STIR sequences at each spinal level. The entire protocol encompassing the cervical, thoracic, and lumbar spine is performed in approximately 40 minutes.
If a fluid collection is identified, dynamic myelography using digital subtraction or CT techniques can precisely locate the leak, allowing percutaneous or surgical treatment. If no extradural fluid collection is present, dynamic myelography can be used to localize CVF. Supplementary MR myelography with intrathecal gadolinium can also be used to localize slowly leaking dural defects and meningeal diverticula.
A large epidural fluid collection with a substantial ventral component almost always suggests an underlying ventral dural defect, often caused by a bony osteophyte. In contrast, epidural fluid collections that have a minimal ventral component but a greater posterolateral component are often caused by ruptured nerve root sleeves or meningeal diverticula [14] (Fig. 3).
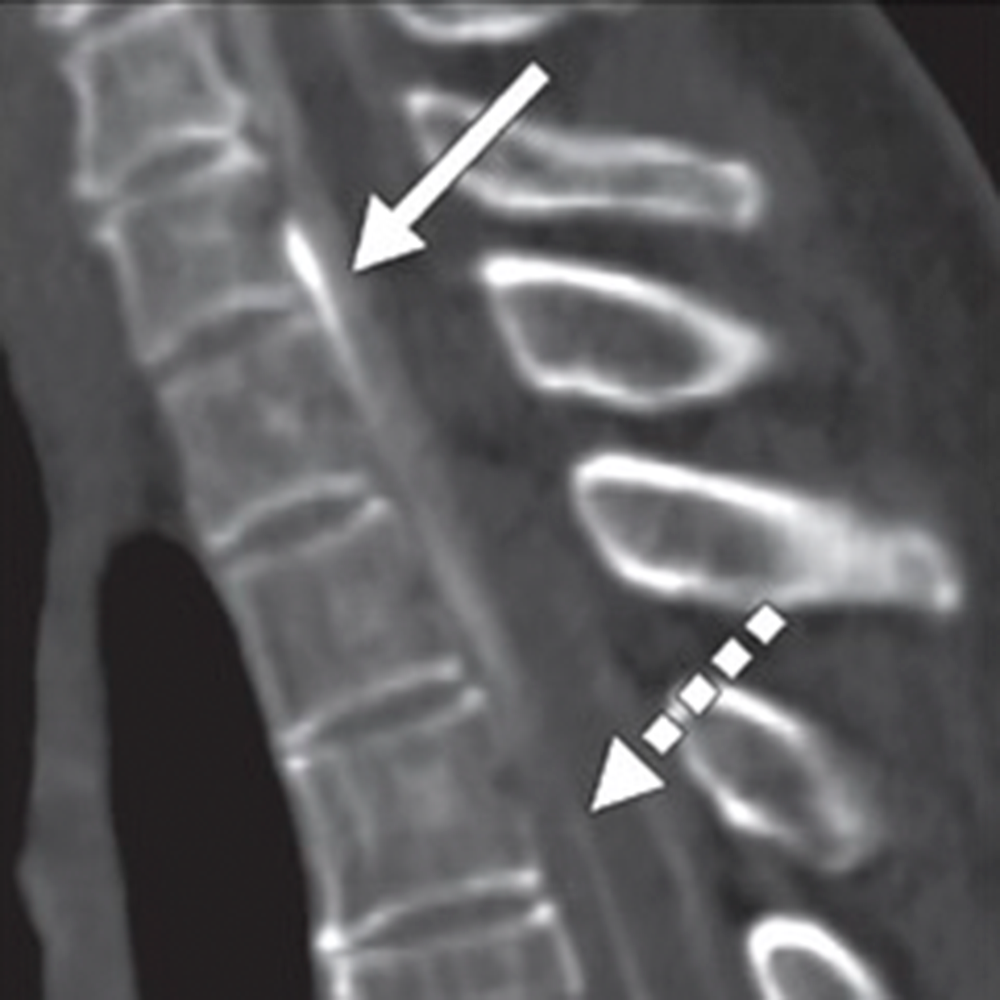
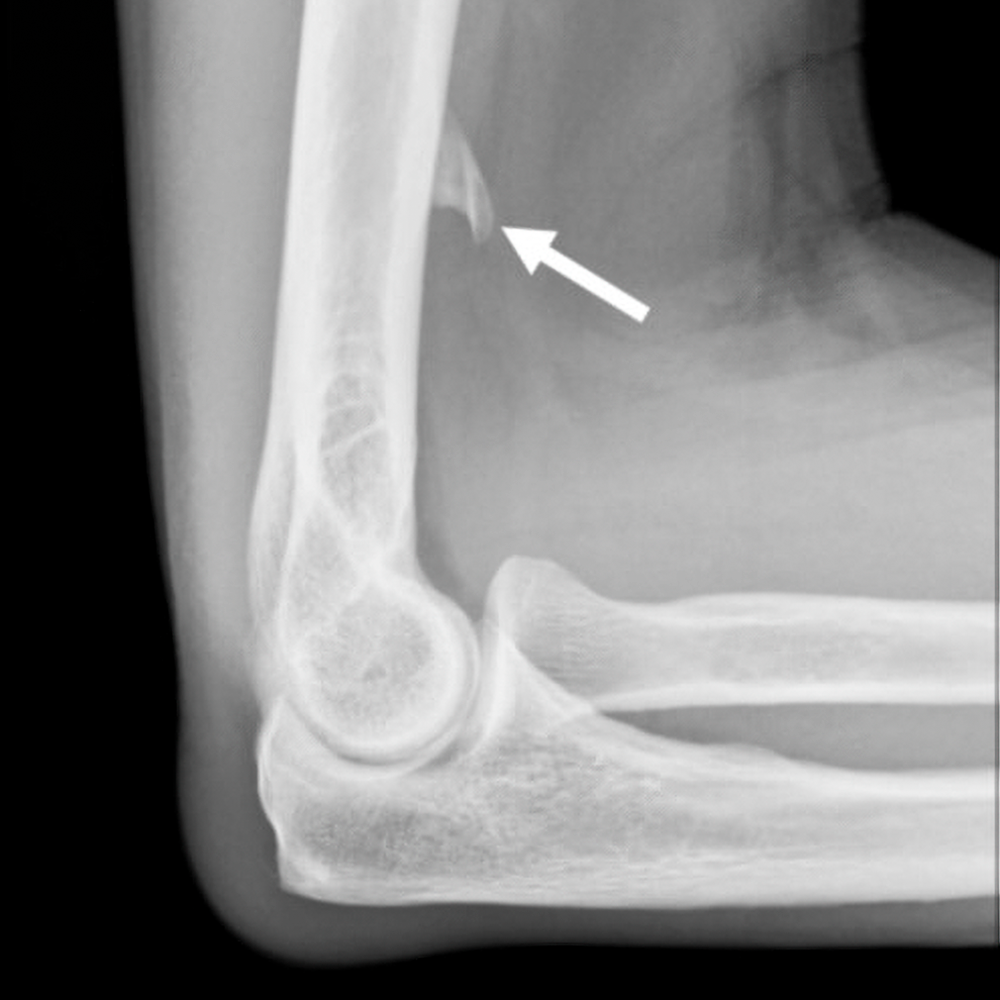
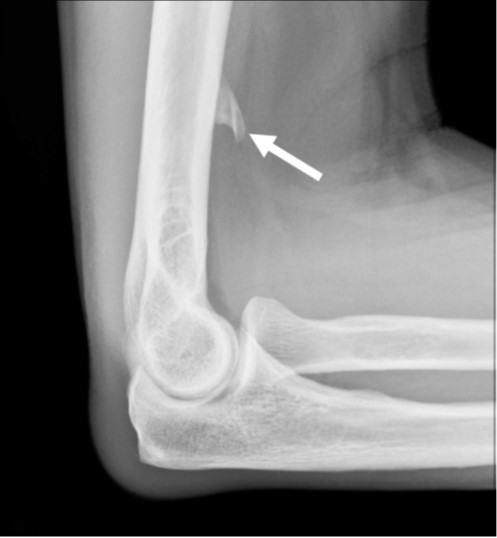
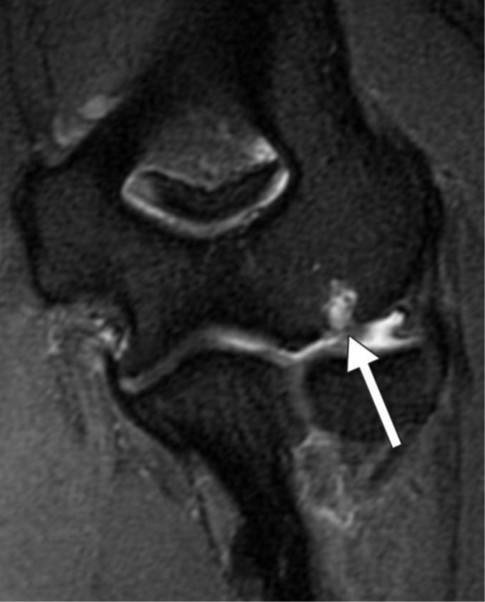
Fig. 3—14-year-old boy with sudden-onset orthostatic headache after playing golf.
A, Sagittal contrast-enhanced T1-weighted MR image shows sagging of brainstem, dural venous sinus, and pituitary engorgement.
B, Axial reconstruction of 3D T2-weighted fat-saturated MR image of spine shows epidural fluid collection confined to dorsal and lateral epidural spaces at T10 level (arrows) without ventral component.
C and D, Axial (C) and coronal (D) slices through left lateral decubitus dynamic CT myelography show pooling of contrast material along lateral epidural space at T10–11 (arrows), arising from ruptured T10 nerve root sleeve.
E, Intraprocedural CT image obtained during CT-guided epidural fibrin glue patching shows percutaneous epidural injection of fibrin and autologous blood. Treatment provided only temporary relief, so patient underwent primary surgical repair.
This distinction is important, because it will dictate patient positioning during dynamic myelography. Thus, the presence and location of epidural fluid collection should be described in the radiology report.
In the absence of epidural fluid collection, the radiologist should report the presence, overall burden, location, and laterality of meningeal diverticula in the spine, because meningeal diverticula are often the nidus of CVF. An asymmetric burden of meningeal diverticula may inform initial patient positioning in dynamic myelography. Additional supplementary findings include any large osteophytes (even in the absence of epidural fluid collection) or enlargement of the thecal sac, which may reflect underlying dural ectasia.
Dynamic Myelography
What distinguishes dynamic from conventional myelography is the timing of imaging and the use of provocative maneuvers such as patient elevation, pressure augmentation, and inspiration to increase conspicuity of CVF [15, 16]. Conventional myelography is often performed in two parts, with the injection of intrathecal iodinated contrast material (often performed under fluoroscopy) followed by whole spine CT after a delay to allow diffusion of contrast material throughout the thecal sac and delineation of the entire subarachnoid compartment. In contrast, dynamic myelography is performed to identify fast, transient egress of CSF into a paraspinal vein laterally. Immediate imaging is obtained after injection of contrast material with the patient in the lateral decubitus position so that contrast material layers along the meningeal diverticula where most CVF tend to arise. Dynamic myelography can be performed with either CT (CTM) or digital subtraction (DSM) techniques. DSM has high temporal resolution but suffers from lack of simultaneous whole spine imaging and is susceptible to motion, superimposition, and breathing artifacts, often requiring general endotracheal anesthesia for technical success. In contrast, dynamic CTM does not require general anesthesia, acquires simultaneous whole spine imaging, and has high spatial localization, allowing optimal treatment planning. DSM may not be available at many institutions; therefore, this chapter discusses dynamic CTM technique in detail. Procedural details of DSM have been published [17].
When an extradural collection is present, the patient will be either prone in case of a suspected dural defect or decubitus when a fast leak from a ruptured meningeal diverticulum or ruptured nerve root axilla is suspected. When CVF is suspected, the patient is placed in the decubitus position.
After intrathecal access is achieved with a spinal needle and iodinated contrast material is injected into the subarachnoid space (5–10 mL of 300 osmolar iodinated contrast material), the patient’s hips are elevated with a foam wedge or inflatable air transfer mattress. When a foam wedge is used, the patient is positioned on the device from the outset, and whole spine CT is performed immediately after injection of contrast material, but because the patient is not in a horizontal position, opening pressure cannot be measured, and thus pressure cannot be reliably manipulated with intrathecal saline infusion. When an inflatable mattress is used, after accessing the subarachnoid space, opening pressure can be measured with a digital manometer and, as long as pressure is normal or low, subsequently augmented with 5-mL aliquots of sterile saline, with repeat measurements after saline infusion to a maximum of approximately 30 cm of water. Subsequently, contrast material is injected, the needle is removed, the device is inflated for approximately 10 seconds, deflated, and then whole spine CT is initiated. In addition, studies have shown that patient inspiration during imaging can increase conspicuity of CVF, presumably lowering central venous pressure and facilitating CSF moving across the fistula [15]. Rehearsing timing of inspiration with the patient before the procedure can be useful to avoid technical failure.
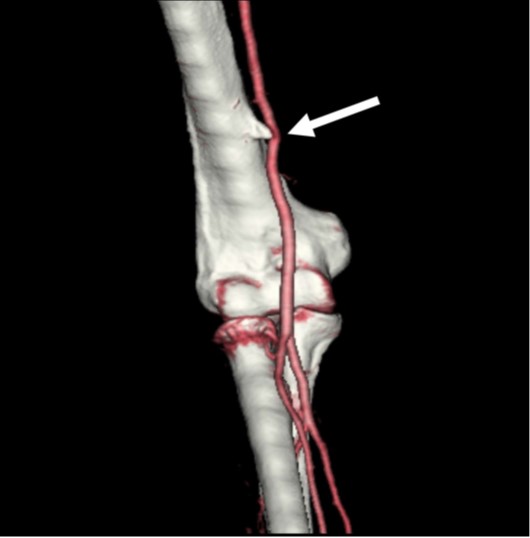
Once whole spine CT is initiated, at least two whole spine acquisitions phases should be performed in succession to maximize temporal resolution. Multiphase acquisition allows scrutinization of paraspinal densities over time, as CVF often fills transiently on a single phase of acquisition (Fig. 4).
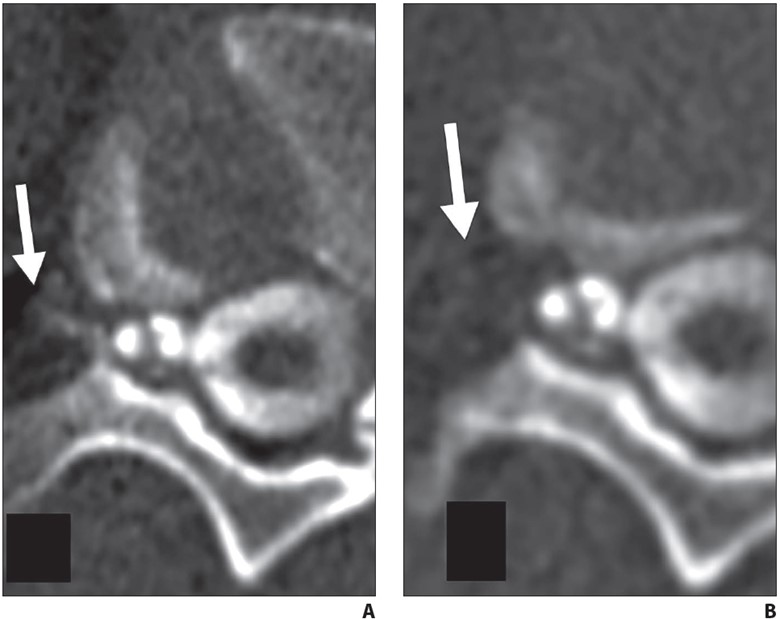
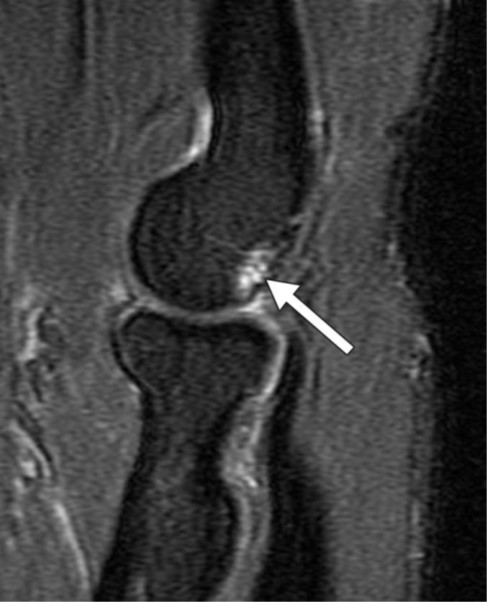
Fig. 4—62-year-old woman with orthostatic headache.
A, Axial early phase image of right lateral decubitus pressure-augmented dynamic CT myelogram shows filling of small paraspinal vein (arrow) lateral to perineural cyst at T11–12 level.
B, Delayed-phase MR image shows contrast material dissipated from vein (arrow), consistent with CSF venous fistula.
Side-by-side comparison of dynamic CT images with preprocedural 3D heavily T2-weighted fat-saturated spine MRI helps distinguish partial filling of perineural cysts from CVF (Fig. 5).
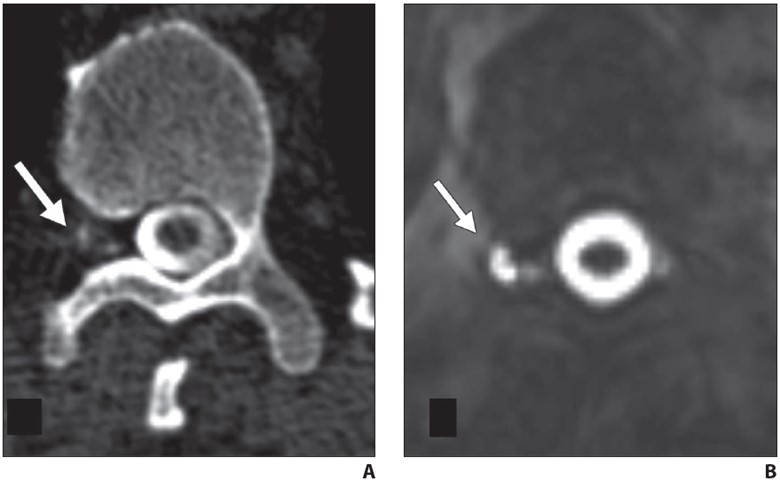
Fig. 5—42-year-old man with orthostatic headaches.
A, Axial decubitus dynamic CT myelogram shows irregular contrast enhancement in right T12-L1 neural foramen (arrow).
B, Axial heavily T2-weighted fat-saturated 3D MR image with CSF leak protocol at same level delineates irregular perineural cyst (arrow), confirming contrast pattern seen in A reflects partial filling of distal portion of perineural cyst rather than CSF venous fistula.
After multiphase whole spine CT is performed in the lateral decubitus position, the patient may be placed in the contralateral decubitus position for one final image, which may reveal a contralateral fistula. Because sensitivity for detecting CVF is highest on the first injection and early phase of imaging, dynamic myelography is traditionally performed in two sessions, one for each laterality. However, some centers have had success leaving the needle in place during patient repositioning, allowing a second injection to complete the contralateral image [18]. Potential concerns with this procedure are excessive needle motion and exacerbation of an iatrogenic dural puncture–related leak. When we perform same-day bilateral decubitus myelography, the needle is either retracted into the ligamentum flavum before repositioning or the needle is removed and a second puncture is performed after repositioning. In patients with underlying connective tissue disease or who may be susceptible to post–dural puncture headache, prophylactic same-day epidural blood or fibrin glue patching at the site of puncture can be performed.
In contrast to CVF localization, pressure augmentation and inspiration maneuvers are not necessary when locating fast leaks from a violation of the ventral or lateral dura. After injection of contrast material into the subarachnoid space, the patient’s hips are elevated and rapid whole spine imaging is performed. Images should be scrutinized for where contrast material first moves from the subarachnoid to the epidural space, filling the epidural fluid collection or lateral epidural space (Fig. 6).
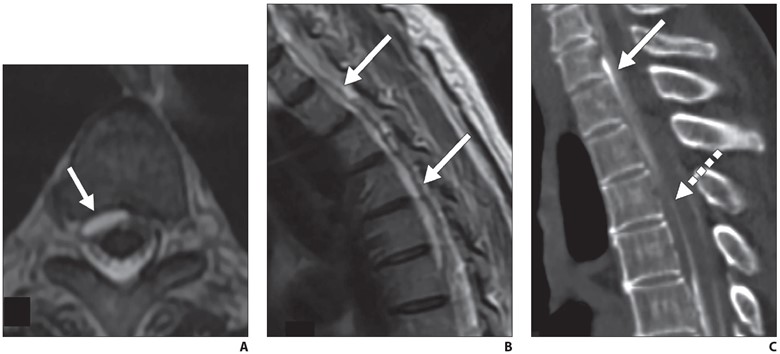
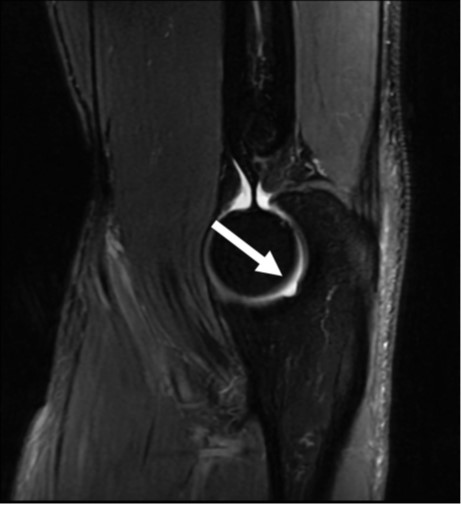
Fig. 6—72-year-old woman with orthostatic headaches.
A and B, Axial (A) and sagittal (B) T2-weighted MR images through thoracic spine show ventral epidural fluid collection extends from T1 to T6 (arrows).
C, Sagittal prone early-phase dynamic myelogram shows filling of ventral epidural fluid collection at C7-T1 level with dense contrast material (solid arrow), while more inferior component of collection (dashed arrow) has not yet filled, suggesting that dural defect is at C7-T1 level.
Regardless of technique, all spinal access should be attempted with a noncutting, pencil point sidehole spinal needle, rather than a traditional cutting spinal needle, which has been shown to minimize the risk of post–dural puncture headache [19].
In cases of high pretest probability (i.e., positive brain MRI and/or highly suspicious clinical symptoms), dynamic myelography may be repeated several times to discover the underlying CVF. Repeated procedures must be weighed against the risk of radiation and multiple dural punctures for a given patient. In patients undergoing repeat dynamic myelography, we may consider concurrent infusion of 0.2 mL intrathecal gadolinium (gadobenate dimeglumine 0.5 M; MultiHance, Bracco) to perform delayed, pressure-augmented MR myelography to potentially reveal a slowly leaking dural defect or meningeal diverticulum. We perform MR myelography using a multiplanar T1-weighted fat-saturated sequence and modified 3-point Dixon fat-suppression technique with 3-mm section thickness with 0.5-mm skip (TR/TE, 475/10; echo train length, 3–4; bandwidth, 50 Hz; matrix size, 320 in frequency-encoding direction and 224 in phase direction). After sagittal full spine acquisition, real-time monitoring is performed to prescribe additional axial and coronal planes of interest to minimize scan time while optimizing image quality.
Treatment Options
CSF Venous Fistula
Percutaneous fibrin glue embolization of CVF is a reasonable first treatment option after a CVF is identified, because it is minimally invasive and can be performed on the same day as dynamic myelography [20]. The technical details of the procedure have been described, but briefly, a 20- or 22-gauge spinal needle is advanced to the origin of the CVF under intermittent CT guidance. When the needle is in the desired position, a test injection of air or dilute contrast material is performed to confirm epidural positioning of the needle. In addition, injection of fibrin glue into the adjacent meningeal diverticulum has been reported to result in technical success without reported chemical meningitis or arachnoiditis [21]; 2–4 mL of fibrin injectate is instilled through the needle, then an image is obtained to observe the injectate spread pattern.
We routinely perform anticipatory fiducial marker placement during fibrin glue injection to facilitate easy relocalization in case of a repeat procedure or subsequent surgery or endovascular treatment. If the first fibrin glue embolization is unsuccessful, a second attempt may be made prior to surgical or endovascular referral. Patients receiving a repeat fibrin injection should be premedicated with 50- mg oral diphenhydramine 30 minutes before the procedure to minimize potential for an allergic reaction. Fibrin glue is a biosynthetic material made from reconstituted human blood components and contains aprotinin, which has been associated with allergic reactions.
At my institution, patients who do not respond to percutaneous fibrin glue embolization are referred for either endovascular embolization or surgical ligation, depending on the patient’s preference and risk tolerance. Surgical ligation of CVF is considered definitive and has been performed the longest of the treatment modalities; however, it is the most invasive treatment option and many patients prefer to avoid surgery if possible. When undergoing endovascular embolization, access to the culprit paraspinal vein is achieved via the azygos or hemiazygos via the internal jugular or femoral vein. After the correct paraspinal vein is catheterized, embolization may be performed with either a liquid embolic system (Onyx, Medtronic) or N-butyl-cyanoacrylate.
Dural Defects and Meningeal Diverticula
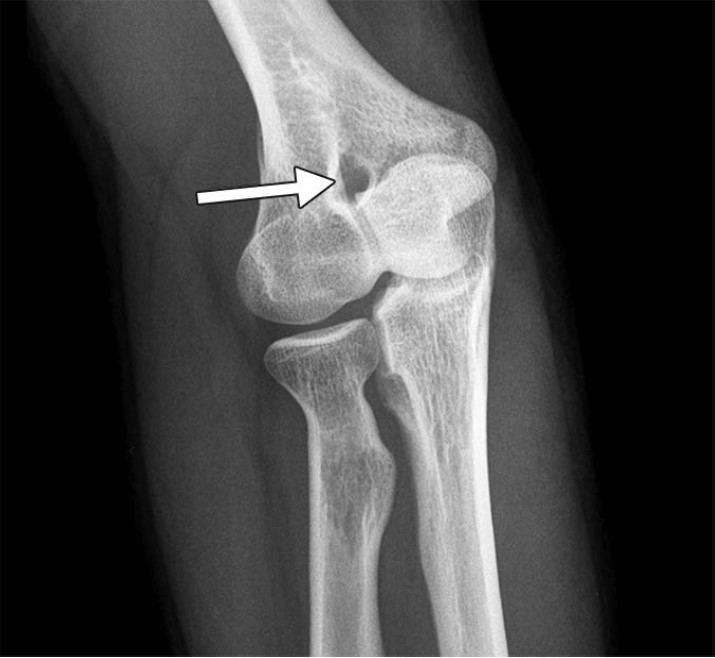
First-line treatment of dural defects and leaking meningeal diverticula is targeted epidural blood and/or fibrin glue patching. The epidural space can be accessed via dorsal interlaminar or transforaminal approaches depending on the type of leak. In dorsal interlaminar approaches, a loss of resistance technique using air can be performed to ideally position the needle in the epidural compartment, whereas in transforaminal approaches, epidural position can be confirmed with injection of a small amount (approximately 0.5 mL) of dilute iodinated contrast material to observe epidural spread of injectate. As many dural defects are ventral, far lateral transforaminal approaches can be used to attempt to access the ventral epidural space (Fig. 7).
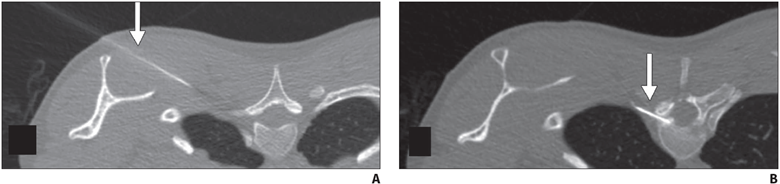
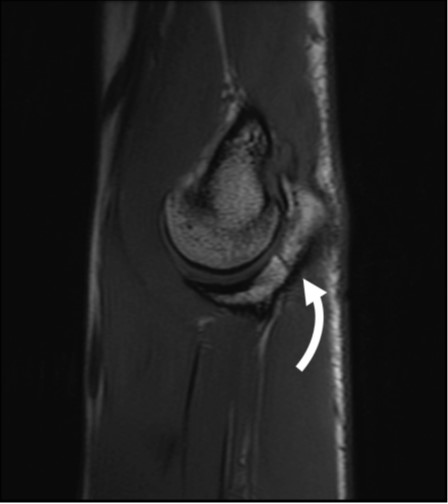
Fig. 7—45-year-old man with orthostatic headaches and confirmed ventral dural defect at T1-2 level.
A and B, Sequential axial CT slices obtained during epidural blood patching show far lateral transmuscular approach using 15-cm spinal needle (arrow) to reach ventral epidural space at T1-2 level.
After treatment, patients should be monitored for resolution of symptoms and repeat imaging performed to ensure resolution of brain findings and epidural fluid collection. Persistent evidence of an epidural fluid collection even in the context of symptom resolution may require surgical treatment, because a chronic dural defect increases the risk of hemosiderosis, which causes its own clinical syndrome and is largely irreversible.
Patients Without a Localized Leak or Fistula
Patients with suspected SIH in whom no leak or fistula is identified on dynamic myelography present a management challenge, particularly if their first-line brain and spine MRI did not show findings of SIH. In these patients, empirical multilevel epidural blood patching is offered for both potential therapeutic and diagnostic purposes. Patients with a strong but temporary clinical response to patching may warrant repeat dynamic myelography. In patients without a clinical response to empirical patching, alternative diagnoses may be considered. My institution has established a multidisciplinary conference consisting of neuroradiologists, neurologists, and neurosurgeons to discuss management of these and other patients to coordinate and optimize care.
SIH is an increasingly recognized, debilitating clinical syndrome for which radiologists have an opportunity to play a key role in diagnosis and treatment. By implementing specialized first-line MRI protocols and dynamic myelography techniques, radiologists can optimize clinical outcomes in this patient population.
References
- Kranz PG, Luetmer PH, Diehn FE, Amrhein TJ, Tanpitukpongse TP, Gray L. Myelographic techniques for the detection of spinal CSF leaks in spontaneous intracranial hypotension. AJR 2016; 206:8–19
- Mokri B. Spontaneous low pressure, low CSF volume headaches: spontaneous CSF leaks. Headache 2013; 53:1034–1053
- Schievink WI, Moser FG, Maya MM. CSF-venous fistula in spontaneous intracranial hypotension. Neurology 2014; 83:472–473
- Callen AL, Timpone VM, Schwertner A, et al. Algorithmic multimodality approach to diagnosis and treatment of spinal CSF leak and venous fistula in patients with spontaneous intracranial hypotension. AJR 2022; 219:292–301
- Mokri B. The Monro-Kellie hypothesis: applications in CSF volume depletion. Neurology 2001; 56:1746–1748
- Schievink WI, Maya MM, Louy C, Moser FG, Tourje J. Diagnostic criteria for spontaneous spinal CSF leaks and intracranial hypotension. AJNR 2008; 29:853–856
- Payer M, Sottas C, Bonvin C. Superficial siderosis of the central nervous system: secondary progression despite successful surgical treatment, mimicking amyotrophic lateral sclerosis—case report and review. Acta Neurochir (Wien) 2010; 152:1411–1416
- Kranz PG, Gray L, Amrhein TJ. Spontaneous intracranial hypotension: 10 myths and misperceptions. Headache 2018; 58:948–959
- D’Antona L, Jaime Merchan MA, Vassiliou A, et al. Clinical presentation, investigation findings, and treatment outcomes of spontaneous intracranial hypotension syndrome: a systematic review and meta-analysis. JAMA Neurol 2021; 78:329–337
- Kranz PG, Amrhein TJ, Choudhury KR, Tanpitukpongse TP, Gray L. Time-dependent changes in dural enhancement associated with spontaneous intracranial hypotension. AJR 2016; 207:1283–1287
- Dobrocky T, Grunder L, Breiding PS, et al. Assessing spinal cerebrospinal fluid leaks in spontaneous intracranial hypotension with a scoring system based on brain magnetic resonance imaging findings. JAMA Neurol 2019; 75:580–587
- Schievink WI, Schwartz MS, Maya MM, Moser FG, Rozen TD. Lack of causal association between spontaneous intracranial hypotension and cranial cerebrospinal fluid leaks. J Neurosurg 2012; 116:749–754
- Magnaes B. Body position and cerebrospinal fluid pressure. Part 2. Clinical studies on orthostatic pressure and the hydrostatic indifferent point. J Neurosurg 1976; 44:698–705
- Madhavan AA, Verdoorn JT, Shlapak DP, et al. Lateral decubitus dynamic CT myelography for fast cerebrospinal fluid leak localization. Neuroradiology 2022; 64:1897–1903
- Amrhein TJ, Gray L, Malinzak MD, Kranz PG. Respiratory phase affects the conspicuity of CSF–venous fistulas in spontaneous intracranial hypotension. AJNR 2020; 41:1754–1756
- Caton MT, Laguna B, Soderlund KA, Dillon WP, Shah VN. Spinal compliance curves: preliminary experience with a new tool for evaluating suspected CSF venous fistulas on CT myelography in patients with spontaneous intracranial hypotension. AJNR 2021; 42:986–996
- Schievink WI, Moser FG, Maya MM, Prasad RS. Digital subtraction myelography for the identification of spontaneous spinal CSF-venous fistulas. J Neurosurg Spine 2016; 24:960–964
- Carlton Jones L, Goadsby PJ. Same-day bilateral decubitus CT myelography for detecting CSF-venous fistulas in spontaneous intracranial hypotension. AJNR 2022; 43:645–648
- Philip JT, Flores MA, Beegle RD, Dodson SC, Messina SA, Murray JV. Rates of epidural blood patch following lumbar puncture comparing atraumatic versus bevel-tip needles stratified for body mass index. AJNR 2022; 43:315–318
- Mamlouk MD, Shen PY, Sedrak MF, Dillon WP. CT-guided fibrin glue occlusion of cerebrospinal fluid-venous fistulas. Radiology 2021; 299:409–418
- Mamlouk MD, Shen PY, Dahlin BC. Headache response after CT-guided fibrin glue occlusion of CSF-venous fistulas. Headache 2022; 62:1007–1018