Published on May 13, 2022

Martin Torriani
Department of Radiology, Division of Musculoskeletal Imaging and Intervention Massachusetts General Hospital and Harvard Medical School
The chest wall includes a variety of osseous, cartilaginous, and musculotendinous structures that are vulnerable to athletic injuries. Lesions involving these structures usually can be divided in two main categories:
- lesions caused by trauma in which an external force causes direct or indirect injury to bones, cartilage, or soft tissue, leading to fractures, dislocations, and soft-tissue contusions;
- lesions caused by mechanical overload from powerful muscle contraction, with or without adequate balance provided by muscle antagonists, leading to myotendinous and muscular lesions and repetitive bone stress changes.
A variety of contact and noncontact sporting activities may have mechanisms that predispose athletes to chest wall injuries. This article summarizes important aspects of athletic chest wall lesions, keeping in mind that many such lesions can occur in nonathletic endeavors, given the similarity of injury mechanisms. General technical aspects for imaging musculoskeletal athletic chest wall injuries are discussed, with most of the focus on CT and MRI, followed by discussions of injuries to the ribs, costochondral cartilage, sternoclavicular joint, and manubriosternal joint. In addition, athletic injuries to select muscles, such as the latissimus dorsi and teres major, are discussed in the context of athletic activities involving throwing.
Technical Considerations
Adequate imaging of chest wall injuries presents technical challenges that are specific to the scanning modality (CT vs MRI) and the targeted structure (rib or costochondral vs soft-tissue lesions). For both CT and MRI, a general recommendation is to perform at least one acquisition with bilateral FOV coverage for comparison purposes. A helpful procedure is to place MRI- or CT-visible skin markers in the area of pain or bracket a zone of discomfort. This allows the radiologist to better focus on specific structures that may be injured but are not clinically or functionally obvious at presentation. Another recommendation regarding MRI includes attempting to minimize respiratory motion by having the patient lie on the affected area (e.g., prone position, if lesion is anterior), which has the combined effect of reducing motion and keeping the structure in contact with the imaging coil. This is, of course, limited by patient comfort, so such decisions may require discretion from the technologist and attending radiologist on a case-by-case basis.
Imaging of bone and costochondral injuries with MRI can be challenging because of the curved anatomy of the chest wall and ribs, which may limit proper visualization of small fractures if nonangled sagittal and coronal planes are used. Obtaining oblique sagittal and/or coronal MR images that are tangential to the abnormality allows identification of the lesion with adequate visualization of the adjacent rib structure and/or costochondral cartilage. Fat suppression is also difficult during chest wall MRI because achieving magnetic field homogeneity often requires the use of more robust techniques, such as STIR pulse sequences. Further, breathing motion and pulsation artifacts may cause image quality degradation, reducing imaging and diagnostic accuracy. The use of fast MRI techniques, such as T2 HASTE, PROPELLER (GE Healthcare), BLADE (Siemens Healthineers), and breath-hold pulse sequences, is desirable, as these approaches mitigate the effect of motion artifacts. On CT, bilateral FOV coverage to compare an affected area with similar contralateral anatomy and the use of thin-slice high-resolution images with bone kernel reconstructions are also recommended. CT allows rapid multiplanar and flexible reconstructions that may be useful when evaluating rib and costochondral fractures, as well as sternoclavicular and manubriosternal joint alignment. Finally, 3D reconstructions are easily obtained from CT images and may prove useful for surgical planning of sternoclavicular joint injuries.
Chest wall muscular injuries are best imaged with MRI, including T1-weighted and T2-weighted fat-suppressed pulse sequences. In the author’s experience, the axial plane is the most important because it provides the best visualization of the pectoralis major, latissimus dorsi, and teres major tendon attachments. Oblique coronal images along the muscle planes can be obtained, though they may provide limited utility for surgical decisions and may be difficult to standardize across multiple sites in an institution. As described for osseous and cartilaginous lesions, the use of fast MRI techniques can help produce images that are less degraded by motion artifact. Bilateral FOV MRI may compromise spatial resolution and should therefore be limited to one to two acquisitions that allow comparison with the unaffected contralateral side.
Sternoclavicular joint injuries are best imaged with CT, especially if clinical signs indicate a posterior dislocation. CT is a prompt imaging method that not only shows the degree of joint malalignment but also evaluates the integrity of upper mediastinum and adjacent great vessels. Manubriosternal joint injuries can be adequately assessed by both MRI and CT, with coronal and sagittal planes being the most adequate to examine for fractures or malalignment.
Ultrasound may prove useful in certain situations. For example, because of the superficial nature of rib and costal cartilage, sonographic detection of fractures is possible and may represent a prompt method to evaluate focal complaints in patients [1]. In this context, the sonographer is able to place the ultrasound probe (preferably using frequencies ≥ 7 MHz) very precisely over the affected area to evaluate for focal osseous or cartilaginous discontinuities. In situations where muscle injuries are suspected, ultrasound is also a reliable imaging method [2]. However, in the author’s experience, it is common for patients with such injuries to present well-developed musculature (e.g., bodybuilders, football players), which may pose a limiting factor for adequate visualization of deep structures, such as tendon attachments. Complementary MRI is required in these cases, with ultrasound serving as a triage tool to be used at the discretion of the medical team. Finally, ultrasound is limited in its capability of assessing malalignments or dislocations of joints, such as the sternoclavicular and manubriosternal joints.
Rib Stress Fractures
Given that ribs are nonweightbearing bones, two main mechanisms can cause rib fractures:
- direct impact from an external source, commonly occurring in football, martial arts, and other contact or extreme sports;
- stress fractures due to muscular contraction.
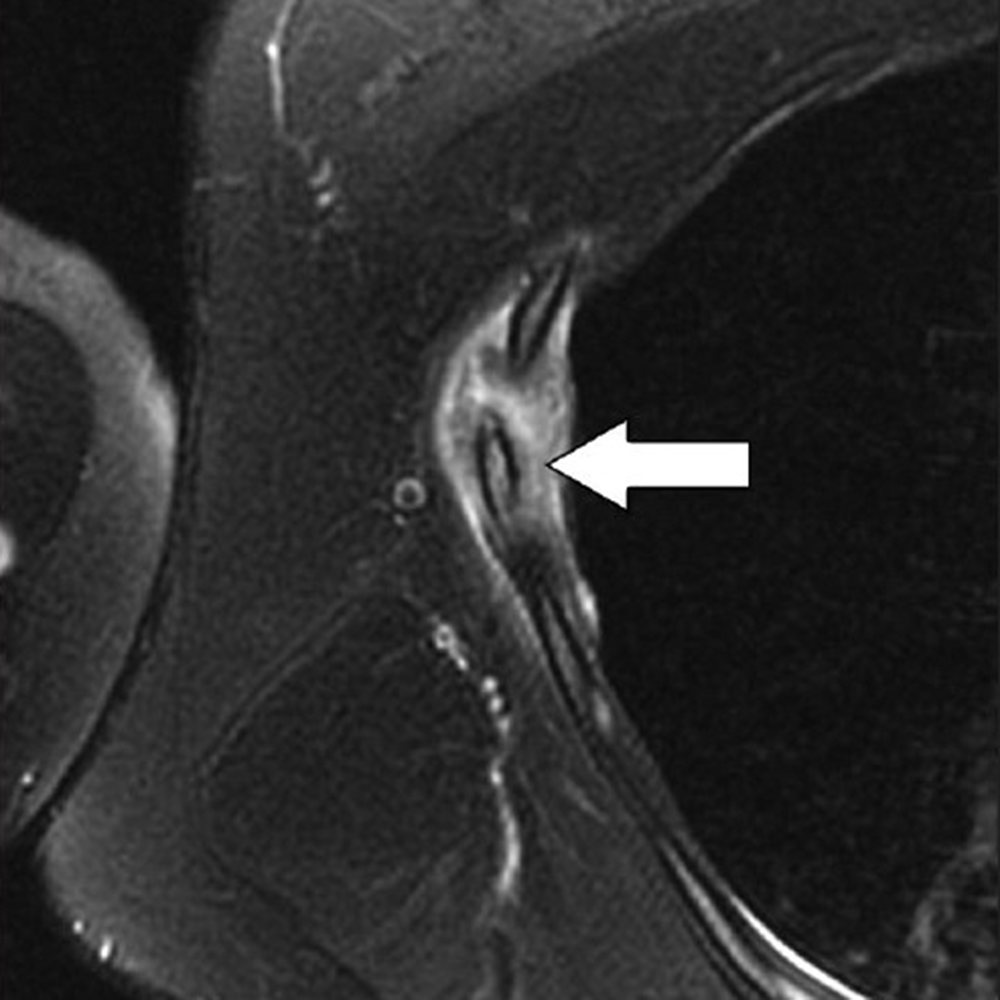
The latter mechanism most often results from strong muscular contraction that is unopposed by a counteracting fatigued muscle. In the case of rib stress fractures, the serratus anterior muscle plays a critical role [3, 4]. The serratus anterior muscle arises from the medial or ventral aspect of the scapula, with multiple slips extending anteriorly that attach to the first through tenth ribs at their middle third. The main role of the serratus anterior muscle is to protract the scapula. Importantly, this action is opposed by the scapular retractors (rhomboid muscles), which stabilize the scapula. In this situation, the serratus anterior muscle will promote an increase in the lateral diameter of the chest cavity. This mechanism can lead to abnormal stress at the middle third of the ribs in sports that involve repetitive contraction of the serratus anterior muscle, such as in elite rowing and swimming. In rowing, stress fractures are relatively common and may affect up to 12% of rowers. The peak contraction of the serratus anterior muscle occurs at the initial phase of the rowing cycle (known as “the catch”), in which the rowing oars are pulled away from the body, causing strong stabilization of the scapula by the rhomboid muscles and serratus anterior muscle. Performed repetitively, this action creates a compressive force vector directed toward the middle thirds of the ribs that can lead to stress fractures, most often affecting the fourth through eighth ribs in up to 86% of cases. If this compression is unopposed by a fatigued serratus anterior muscle, a repetitive stress injury may occur [3, 4]. Stress fractures also affect novice golfers who are still developing their technique and often strike the ground during swing strokes. Frequent ground impact force due to repetitive inaccurate strokes is thought to be transmitted to the chest wall on the leading side, causing stress fractures [5]. Additional sports in which rib stress fractures have been described include baseball, lacrosse, weightlifting, ballet, and gymnastics. MRI findings of rib stress fractures are similar to those seen in the extremities, including a well-defined linear area of low signal intensity surrounded by marrow edema, with the best contrast noted in T2- weighted fat-suppressed images (Fig. 1).

Edema in the surrounding intercostal muscles and extrapleural space is often noted [6]. As mentioned earlier, such fractures may be better depicted on oblique sagittal or coronal images that are tangential to the fracture and provide context, better showing rib portions adjacent to the fracture itself. Healing of rib stress fractures can be monitored by MRI, showing progressive improvement of marrow edema and bony bridging; however, clinical symptoms may persist even though the fracture appears healed on imaging.
Stress fractures affecting the first rib result from a different proposed mechanism that is a function of the rib’s anatomic features. The serratus anterior muscle attaches to the first rib’s lateral edge at its middle third, whereas the anterior and middle scalene muscles attach to the medial edge at the same level. The anterior scalene muscle attaches at a well-defined bony protuberance, behind which is a biomechanically weak zone of the first rib. This area, through which the subclavian vessels and brachial plexus travel, has been dubbed the “Achilles heel” of the first rib [7]. First rib fractures occur more commonly in throwing athletes, such as baseball players. Throughout the throwing cycle, the most prominent eccentric muscular contraction of the serratus anterior muscle occurs at the arm cocking stage, during which the scapula moves posteriorly, leading to strong opposition by the serratus anterior muscle. This motion, performed repetitively in high-performance athletes, can place undue mechanical stress on the weak zone of the first rib, leading to stress fractures. Such fractures can be treated conservatively but may take a long time to heal (6–12 months) and, if nonunion occurs with a large bone callus, may lead to symptoms of thoracic outlet syndrome. Additional activities for which first rib fractures have been described include jive dancing, basketball, tennis, and weightlifting. First rib fractures can be depicted adequately on MRI, though most studies on such fractures have used CT and occasionally nuclear medicine bone scans [7].
Costal Cartilage Injury
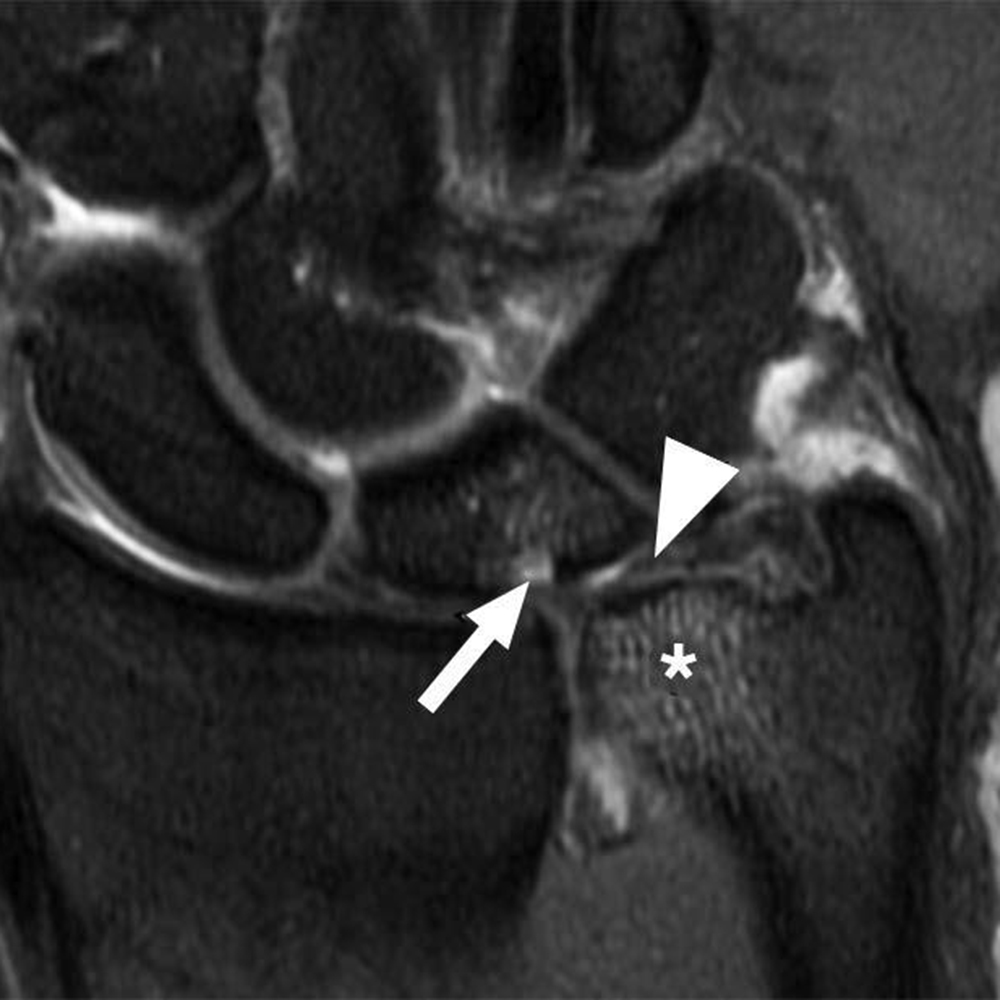
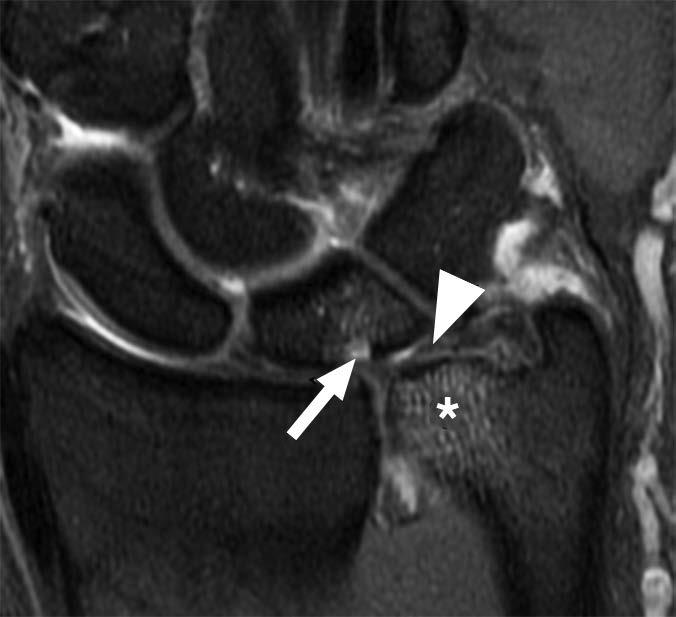
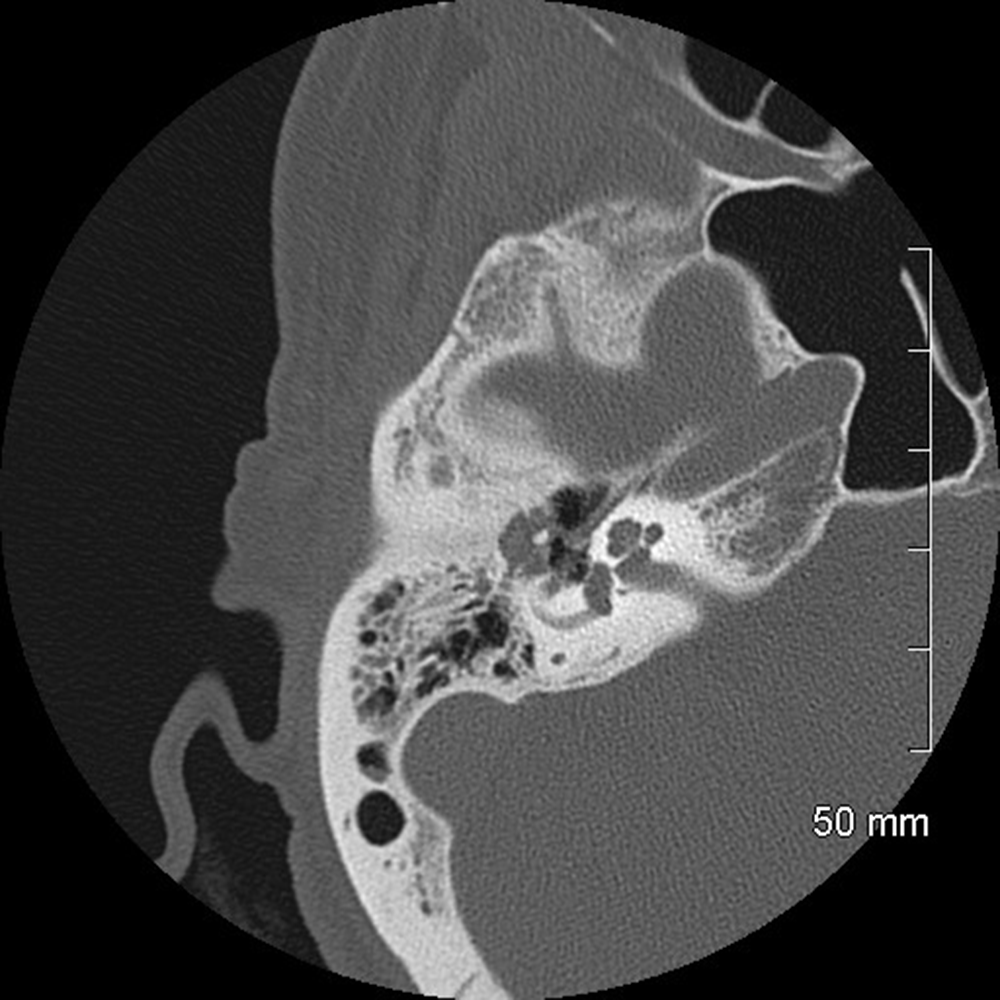
Areas that may be affected by costal cartilage injury include the sternochondral junction, midsubstance of the costal cartilage, and costochondral junction. Most commonly, such injuries affect the relatively immobile upper rib cage (first to third costal cartilages) and are due to rotation injury and/or excessive axial loading (such as in weightlifting). Lesions affecting the lower rib cage (fourth through eighth costal cartilages) typically result from direct impacts, such as seen in contact sports (hockey, football, rugby, and martial arts) [8]. An important anatomic feature of costal cartilage is the presence of a central cavity that is best seen on CT images as a zone of lower attenuation tracking along the structure [9]. A predominance of blood vessels, loose connective tissue, macrophages, and fat exist within this area. Although this zone resembles a bone marrow cavity, no marrow cells or lineage is present. Anatomic studies suggest this central channel in costal cartilage is more akin to nutrient channels because of the dominant feature of high vascularity. The presence of a dense vascular network in this portion of costal cartilage predisposes to significant bleeding in the event of fractures. A common feature in costal cartilage fractures is the presence of an associated hematoma of the chest wall, which can lead to clinician suspicion of neoplastic masses. Scrutiny of the area using thin-slice CT may reveal a linear discontinuity of the costal cartilage adjacent to the suspected mass, increasing the likelihood of a trauma-related cause. Patient history may help direct the radiologist, as a traumatic event related to lifting or direct impact may have occurred, possibly resulting in subsequent pain and a local mass (Fig. 2).

MRI provides excellent contrast to show costal cartilage fractures, which will appear as linear areas of high signal intensity against a background of low-signal-intensity costal cartilage [8]. Although MRI has superb spatial resolution and contrast, its relatively higher cost and susceptibility to respiratory motion artifacts decrease its effectiveness, when compared with CT. Ultrasound may also prove useful, given the superficial location of costochondral structures, and may show a focal stepoff at the fracture area [1]. As previously mentioned, costal cartilage injuries may occur at the sternochondral or costochondral junctions. At these locations, chondral and adjacent bone marrow edema may be present on MRI and represent the dominant findings related to traumatic injury. Treatment of costal cartilage injuries include NSAIDs, taping and immobilization belts, and rest for 2–3 weeks. In certain situations, such as treatment of professional athletes, pain can be mitigated by local injections of anesthetic [8]. An important differential diagnosis to consider when entertaining the possibility of a costochondral injury is Tietze syndrome. In this rare entity, inflammation of the sternochondral junction (the most common symptom) can occur spontaneously and lead to focal pain without any specific initiating event. The cause of Tietze syndrome is uncertain, and the condition usually affects younger patients (< 40 years old). On MRI, this entity may show edematous change surrounding the sternochondral junction, which is best seen on fat-suppressed T2-weighted images [10]. CT may not provide enough tissue contrast for adequate detection. Given that Tietze syndrome is usually self-limited, its treatment includes rest, NSAIDs, and local anesthetic injections in refractory cases.
Sternoclavicular Joint
Athletic injuries to the sternoclavicular joint may lead to anterior dislocation (more common and less concerning) or posterior dislocation [11, 12]. The latter phenomenon is less frequent, but more concerning, because the clavicular head has the potential to exert mass effect on the great vessels, trachea, esophagus, and recurrent laryngeal nerve (causing vocal cord palsy). Posterior dislocations require 50% more force to occur, compared with anterior dislocations. This is because the capsular structure of the sternoclavicular joint is more robust posteriorly [13]. Posterior sternoclavicular joint dislocations are most commonly related to direct-force injuries seen in football, rugby, martial arts, and motor vehicle accidents (typically motorcycle accidents). Sternoclavicular dislocations can be imaged by CT and/or MRI, both of which may show malalignment at the joint with associated fractures or hematomas from capsular disruption. In the case of posterior dislocations, CT provides excellent delineation of the joint alignment, and use of IV contrast material allows assessment of the integrity of adjacent vascular structures. Alignment of mediastinal structures and possible hematomas are also best seen on CT. The initial treatment of posterior sternoclavicular dislocations includes attempted closed reduction, which is usually performed under sedation as an urgent procedure. If this procedure fails to realign the joint, or if the joint remains unstable, a surgical approach involving open reduction and internal fixation may be necessary.
Manubriosternal Joint
The manubriosternal joint is rarely involved in athletic injuries. Usual mechanisms of injury are classified as either type 1, in which an anteroposterior force is exerted on the sternal body, which moves posteriorly relative to the manubrium, or type 2, in which the anteroposterior force is exerted against the manubrium [14]. The latter mechanism is usually related to forces being transmitted through the arms to the clavicle and sternoclavicular joints, displacing the manubrium posteriorly. In the author’s experience, such lesions are rare and show marrow edema surrounding the manubriosternal joint on MRI, suggesting local bone contusions. Both sagittal and coronal images through the sternum are optimal for this visualization.
Latissimus Dorsi and Teres Major
Injuries to the latissimus dorsi and teres major muscles are most commonly seen in throwing athletes [15–18]. The latissimus dorsi muscle has a broad origin that includes, among others, multiple spinous processes of the thoracolumbar region and iliac crest; the muscle then attaches at the floor of the intertubercular groove of the humerus. The teres major muscle originates at the posterior aspect of the inferior angle of the scapula and inserts at the medial ridge of the intertubercular sulcus of the humerus. In cadaver studies, the latissimus dorsi and teres major tendon attachments were seen as a single structure in up to 83% of cases [15–18]. In the remaining cases, separate tendons attaching to the humerus were visible on axial images. One important action of these muscles is to powerfully adduct, extend, and internally rotate the humerus. During the throwing motion, the latissimus dorsi and teres major muscles are activated as a unit, with maximum levels of eccentric contraction during the arm cocking and arm deceleration stages of the throwing cycle [15–18]. Lesions of the latissimus dorsi and teres major may present along the usual spectrum of myotendinous strain injuries up to avulsions at the humeral attachments. For this reason, MRI is the preferred imaging method, as it can characterize the full gamut of possible lesions, including those affecting the more dorsal portions of the latissimus dorsi muscle. In this situation, the location of the patient’s symptoms should be considered to ensure adequate coverage on imaging. Because the latissimus dorsi covers a large area of the lateral and posterior chest wall, large FOVs may be required, especially if bilateral imaging is being performed. Another important technical point is to ensure that axial images through the humerus adequately cover the footprints of latissimus dorsi, teres major, and pectoralis major tendons, which are usually not fully viewed during standard shoulder imaging protocols. This may require obtaining axial images that extend caudally to approximately the middle third of the humerus to guarantee such coverage in all patients. Regarding treatment, avulsions at the humeral attachment, which have been described in athletes involved in waterskiing, golf, tennis, and bodybuilding, usually require surgical intervention to reattach the tendons [18]. On the other hand, most myotendinous lesions of latissimus dorsi and teres major muscles are treated conservatively.
Imaging of athletic chest wall injuries should be performed while taking into account anatomic considerations and the advantages of specific imaging methods, including CT, MRI, and ultrasound. Because these injuries are likely to comprise a relatively smaller volume of cases at a given imaging facility, predefined workflows are important to tackle targeted anatomic locations. This may require close communication between scheduling staff, technologists, and attending radiologists, who can tailor protocols to obtain the best images of athletic chest wall injuries.
References
- Malghem J, Vande Berg B, Lecouvet F, Maldague B. Costal cartilage fractures as revealed on CT and sonography. AJR 2001; 176:429–432
- Chiavaras MM, Jacobson JA, Smith J, Dahm DL. Pectoralis major tears: anatomy, classification, and diagnosis with ultrasound and MR imaging. Skeletal Radiol 2015; 44:157–164
- Warden SJ, Gutschlag FR, Wajswelner H, Crossley KM. Aetiology of rib stress fractures in rowers. Sports Med 2002; 32:819–836
- McDonnell LK, Hume PA, Nolte V. Rib stress fractures among rowers. Sports Med 2011; 41:883–901
- Lord MJ, Ha KI, Song KS. Stress fractures of the ribs in golfers. Am J Sports Med 1994; 24:118–122
- Taimela S, Kujala UM, Orava S. Two consecutive rib stress fractures in a female competitive swimmer. Clin J Sport Med 1995; 5:254–256; discussion, 257
- Coris EE. First rib stress fractures in throwing athletes. Am J Sports Med 2005; 33:1400–1404
- Subhas N, Kline MJ, Moskal MJ, White LM, Recht MP. MRI evaluation of costal cartilage injuries. AJR 2008; 191:129–132
- Lee S, Choi YW, Jeon SC. Low attenuation areas in normal costal cartilages on CT: clinical implication and correlation with histology. Clin Anat 2012; 25:483–488
- Volterrani L, Mazzei MA, Giordano N, Nuti R, Ga- leazzi M, Fioravanti A. Magnetic resonance imaging in Tietze’s syndrome. Clin Exp Rheumatol 2008; 26:848–853
- Mirza AH, Alam K, Ali A. Posterior sternoclavicular dislocation in a rugby player as a cause of silent vascular compromise: a case report. Br J Sports Med 2005; 39:e28
- Galanis N, Anastasiadis P, Grigoropoulou F, Kirkos J, Kapetanos G. Judo-related traumatic posterior sternoclavicular joint dislocation in a child. Clin J Sport Med 2014; 24:271–273
- Spencer EE, Kuhn JE, Huston LJ, Carpenter JE, Hughes RE. Ligamentous restraints to anterior and posterior translation of the sternoclavicular joint. J Shoulder Elbow Surg 2002; 11:43–47
- Hayashi D, Roemer FW, Kohler R, Guermazi A, Gebers C, De Villiers R. Thoracic injuries in professional rugby players: mechanisms of injury and imaging characteristics. Br J Sports Med 2014; 48:1097–1101
- Schickendantz MS, Kaar SG, Meister K, Lund P, Beverley L. Latissimus dorsi and teres major tears in professional baseball pitchers: a case series. Am J Sports Med 2009; 37:2016–2020
- Ellman MB, Yanke A, Juhan T, et al. Open repair of an acute latissimus tendon avulsion in a Major League Baseball pitcher. J Shoulder Elbow Surg 2013; 22:e19–e23
- Escamilla RF, Andrews JR. Shoulder muscle recruitment patterns and related biomechanics during upper extremity sports. Sports Med 2009; 39:569–590
- Cox EM, McKay SD, Wolf BR. Subacute repair of latissimus dorsi tendon avulsion in the recreational athlete: two-year outcomes of 2 cases. J Shoulder Elb Surg Am 2010; 19:e16–e19